Current location: Home > NEWS > Industry news
NEWS
PRODUCTS
2026 NCCN Breast cancer Guideline Update-Precision Treatment adds a new direction
News source: Release time:[2026-01-28]
As the “number one threat” to women's health, the diagnosis and treatment of breast cancer have always been advancing rapidly in the direction of precision and individualization. On January 16, 2026, NCCN officially released the "Guidelines for the Diagnosis and Treatment of Breast Cancer (2026. V1 edition)", which have brought key updates in many fields such as molecular detection, targeted therapy, and immunotherapy, and provide clearer guidelines for clinical decision - making.

01. Surgery and radiotherapy: the exemption strategy is clearer
· Expansion of the exempted population for sentinel lymph node biopsy (SLNB):Based on SOUND and INSEMA trial data, patients who are >50 years old, postmenopausal, cT1N0 (negative axillary ultrasound), HR+/HER2-, Grade 1-2, and agree to receive whole breast radiotherapy and endocrine therapy may consider omitting SLNB. Caution is required regarding the histological types of lobular cancer.
· Patients with low-risk DCIS may be exempted from radiotherapy:Patients with “low-risk” ductal carcinoma in situ (DCIS) who meet the RTOG 9804 standard (screening discovery, Grade 1-2, size ≤62.5px, incision ≥3mm) can consider endocrine therapy only and omit radiotherapy.
02. Adjuvant therapy: New drugs, new evidence, new options
· HER2+ early - stage breast cancer: The results of the long - term follow - up of the APHINITY trial (median 11.3 years) confirmed that adding Pertuzumab to Trastuzumab on the basis of Trastuzumab + chemotherapy can continue to bring recurrence prevention benefits.
· A new choice for patients with high risk of recurrence of HER2+: For high - risk patients who still have residual lesions (non - pCR) after neoadjuvant treatment (such as being inoperable at first or having axillary lymph nodes still positive after neoadjuvant treatment), T - DxD has become a category 1 recommended choice for adjuvant therapy.
03. Late - stage treatment: A huge change in the pattern of targeted therapy
· The frontline status of ADC drugs has been upgraded:
1) Sacituzumab Govitecan combined with Pembrolizumab has become a category 2A priority recommendation for the first - line treatment of triple - negative breast cancer (TNBC).
2) T - DxD is added to the first - line option as another recommended option for HR+/HER2 - advanced breast cancer.
· Update to the targeted combination regimen: For HER2 - advanced breast cancer with PIK3CA mutations, the Fulvestrant/Inavolisib/Palbociclib combination regimen has been included in the recommendation. The combination of Imlunestrant + Abemaciclib has also become an option for follow - up treatment.
This update once again strengthens the concept of “detection first, accurate decision - making”. The following is a summary of the key molecular markers that must be paid attention to at all stages of clinical trials:
01. Core markers of diagnosis and prognosis stratification
· Hormone receptors (HR): including ER (estrogen receptor) and PR (progesterone receptor). The guidelines emphasize that even if ER expression is low (1% - 10%), the potential benefits of endocrine therapy should be discussed with patients, but the risks and benefits need to be individually weighed.
· HER2 status: Use the ASCO/CAP guidelines for IHC and ISH testing. Special attention is paid to the determination of HER2 - low (IHC 1+ or 2+/ISH -), which is directly related to the use of ADC drugs.
· Ki - 67 index: When high - risk HR+/HER2 - patients consider using Ribociclib, Ki - 67≥20% is one of the decision - making factors.
· Multi - gene expression profile detection: used for prognostic assessment and prediction of chemotherapy benefit in early HR+/HER2 - patients.
1) 21 Genetic test (Oncotype DX): For patients with pN0 and pN1 (1 - 3 positive lymph nodes), it is still a preferred tool recommended by the NCCN team to guide chemotherapy decision - making.
2) Other testing tools: such as 70 genes (MammaPrint), 50 genes (Prosigna), 12 genes (EndoPredict), breast cancer index (BCI), etc., also provide important prognostic information, especially in assessing the risk of long - term recurrence and prolonging the benefits of endocrine therapy (such as BCI).
02. Genetic risk assessment markers
· BRCA1/2 embryonic mutations: Not only affect genetic risk assessment but also are directly related to treatment options. Carriers can consider using Olaparib in the neoadjuvant/adjuvant phase. At the same time, it is also one of the exclusion criteria for breast preservation radiotherapy (APBI/PBI).
· Other breast cancer susceptibility genes: such as ATM (homozygous mutations are absolute contraindications for breast preservation radiotherapy), TP53 (Li - Fraumeni syndrome), PALB2, etc., need to be evaluated based on family history and personal history to guide surgery and screening strategies.
03. Late - stage treatment target detection
· PIK3CA mutation: It is a prerequisite for the use of Alpelisib or Inavolisib combined regimen for HR+/HER2 - advanced breast cancer.
· ESR1 mutation: Detected in patients with advanced HR+ who have received endocrine therapy, it is related to sensitivity to novel oral SERDS (such as Imlunestrant) and combination regimens. The guidelines recommend that ctDNA testing be given priority after the disease progresses.
· NTRK fusion, MSI - H/dMMR, TMB - H: As a target of “unlimited cancer types”, it is worth testing after standard treatment fails.
· Comprehensive genetic analysis: The guidelines recommend multi - gene panel testing (including somatic cells and embryonic lines) for recurrent/metastatic breast cancer to systematically find targeted treatment opportunities and eligibility for clinical trials.
04. New markers and detection technologies
· Molecular residual lesion monitoring: After neoadjuvant therapy, the assessment of tumor residual load is the standard. At the same time, ctDNA monitoring has shown the potential to predict recurrence in high - risk patients, although more evidence is needed for routine recommendation in the guidelines.
SpaceGen is deeply engaged in the field of precision tumor detection and is committed to providing comprehensive detection kit solutions for personalized precision medical detection of tumors. For breast cancer - related targets, comprehensive coverage and accurate detection can be achieved, effectively reducing missed tests, and helping to open a new chapter in the precise treatment of breast cancer.
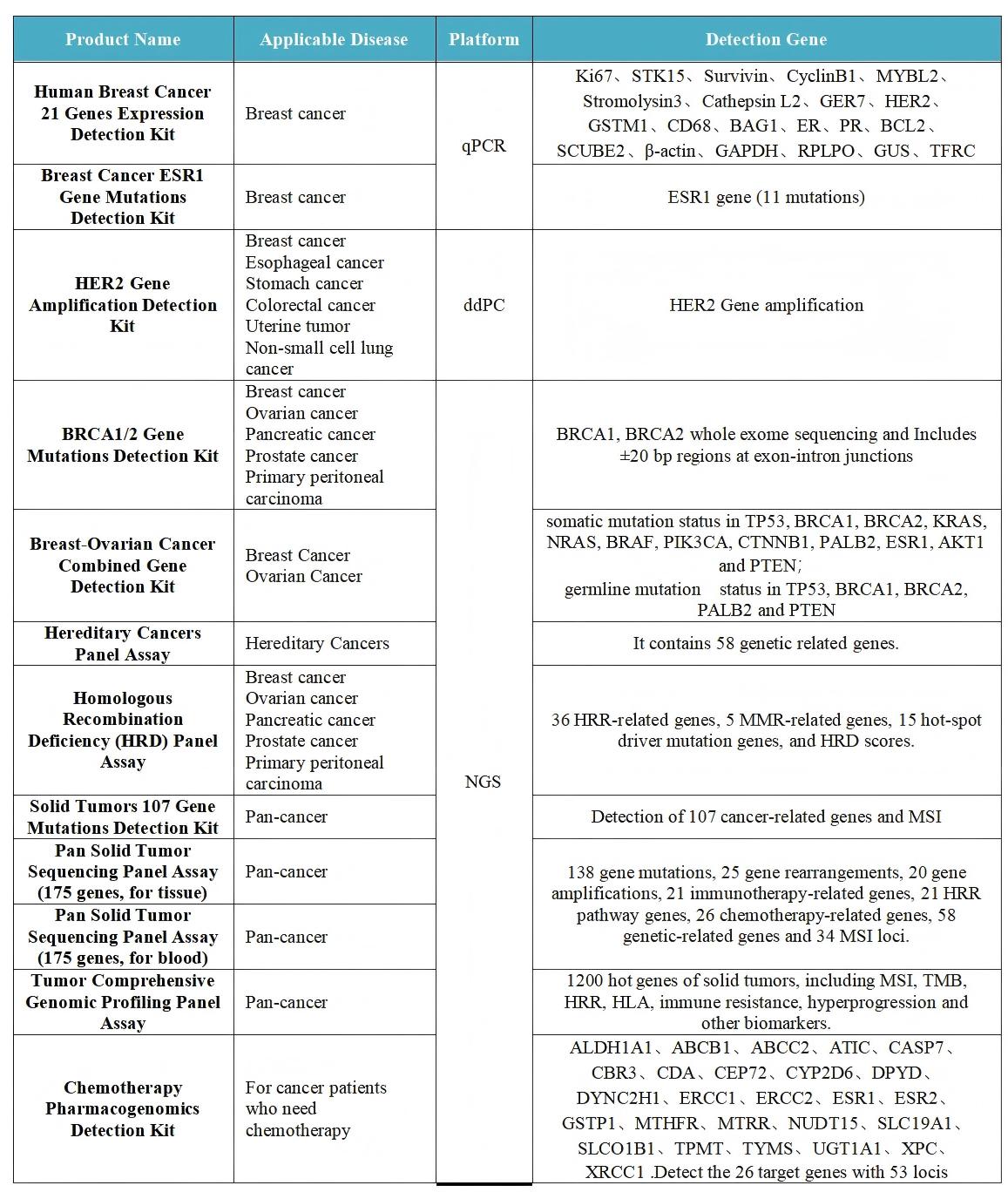
The NCCN Breast Cancer Guidelines 2026, Version 1 update, represents another significant advancement for precision medicine in the field of breast cancer. From the optimization and enhancement of early - stage treatment to targeted breakthroughs in late - stage treatment, every step is closely associated with the guidance of precise molecular detection.
References
NCCN Clinical Practice Guidelines in Oncology: Breast Cancer 2026, V1
Statement: This article is solely for sharing purposes. If it involves copyright or other related issues, please contact us promptly, and we will make corrections as soon as possible. Thank you!