Current location: Home > NEWS > Industry news
NEWS
PRODUCTS
What is the most promising strategy to overcome PARP inhibitor resistance in ovarian cancer?
News source: Release time:[2025-11-24]
01. Overview
Ovarian cancer is the deadliest form of gynecological cancer. Despite the introduction of Bevacizumab, the standard chemotherapy regimen has largely remained the same, and the majority of patients experience a relapse within the first two years following diagnosis. However, clinical practice outcomes suggest that the clinical benefits of PARP inhibitor therapy are swiftly altering the treatment options for many individuals with ovarian cancer.
Clinical trials have shown that platinum-based chemotherapy is beneficial as a maintenance treatment after achieving an initial complete response (CR) or partial response (PR) for both primary and recurrent ovarian cancer. This is partly due to the high prevalence of tumors with homologous recombination defects (HRD), which are found in approximately 50% of all ovarian cancers. Deficiency in BRCA1/2 accounts for about 20% of these cases [1]. This forms the molecular basis and significant clinical benefits of PARP inhibitors in patients with BRCA1/2 mutations and in those with HR-deficient tumors. Although PARP inhibitors also demonstrated a significant prolongation of progression-free survival (PFS) in patients with wild-type BRCA1/2 and HR-related tumors, this clinical benefit was less pronounced than in the other subgroups mentioned above.
02. Understanding the potential mechanisms of resistance is crucial for enhancing the clinical application of PARP inhibition in ovarian cancer
Although relevant clinical trials have shown promising results by significantly improving progression-free survival (PFS), key issues remain. It is clear that most patients eventually develop resistance to PARP inhibitors (PARPi). The duration of resistance may depend on the uniqueness of the efficacy of PARPi, namely, the BRCA mutation and the homologous recombination deficiency (HRD) status. One can speculate that the complexity of HRD in tumors is the basis for the development of drug resistance. This means that resistance to PARPi may develop more rapidly if PARPi does not need to counteract the (relatively) complex mechanisms. This hypothesis is based on clinical observations that patients with tumors classified as "HR-normal" benefit less from PARPi after platinum-based chemotherapy than patients with HR-deficient or BRCA1/2 mutant tumors. Genomic analysis of long-term and short-term responders to Olaparib revealed that Olaparib responses were associated with BRCA1/2 mutations. It is further speculated that the potential types of BRCA mutations may provide a more accurate prediction of long-term responders.
Similarly, more accurate HRD testing may provide a better understanding of the factors that contribute to a sustained clinical response to PARP inhibition. On the one hand, the potential method for identifying new biomarkers is to reveal the potential drug resistance mechanism in patients who fail to respond to PARP inhibition. On the other hand, it will be essential to identify long-term survivors of BRCA wild-type (BRCAwt) and HR-normal tumors treated with PARPi and characterize potential biomarkers that may predict this abnormal response. This exciting pathway will also shed light on potential strategies to improve the efficacy of PARPi.
03. Mechanisms of Resistance and Strategies to Improve Efficacy
Numerous potential mechanisms of PARP inhibitor (PARPi) resistance have been investigated. The strongest theoretical foundation for the clinical development of PARPis stems from the responses observed in BRCA1/2-deficient (and homologous recombination (HR)-deficient) cells. Consequently, an intriguing resistance mechanism involves secondary somatic reversions of the BRCA1/2 gene in tumor cells, which essentially restore HR function. This phenomenon may be attributed to tumor heterogeneity and clonal expansion induced by chemotherapy, often referred to as "Darwinian escape."
A study comparing primary ovarian cancer with recurrent ovarian cancer found that 13 out of 46 recurrent high-grade serous ovarian cancer (HGSOC) cases had secondary mutations, and two out of 64 primary HGSOC cases were carriers of germline BRCA1/2 mutations [3]. This trend was more pronounced in platinum-resistant and platinum-sensitive ovarian cancers, where nearly 50% of platinum-resistant ovarian cancers (12 out of 26) exhibited BRCA1/2 reversion. The secondary reversion mutation in BRCA1/2 has also been associated with PARPi resistance in ovarian and other cancers. Most intriguingly, secondary revertant mutations in the BRCA1/2 allele have been detected by analyzing circulating free DNA (cfDNA) in patients with prostate cancer. This provides a non-invasive method to more accurately predict the efficacy of platinum-based chemotherapy and/or PARPi therapy [4]. Other mechanisms affecting gene expression include BRCA1/2 hypermethylation. RAD51, a crucial component of the HR pathway, may also undergo secondary reverse mutations, potentially leading to PARPi resistance in platinum-sensitive HGSOC patients treated with Rucaparib.
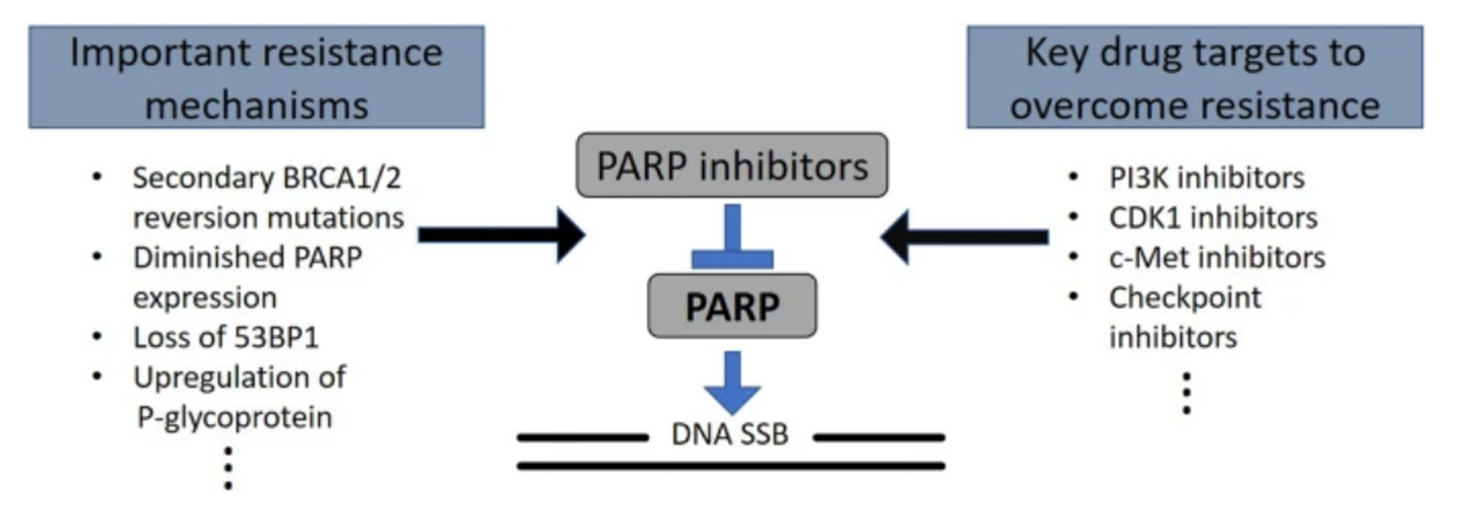
Drug resistance mechanisms and potential targets of combination therapy. A summary of resistance mechanisms and potential drug targets, along with corresponding inhibitors for use with PARP inhibitors (PARPi).
However, the efficacy of PARPi may also be influenced by a BRCA-independent mechanism for homologous recombination (HR) recovery. TP53 binding protein 1 (53BP1) is another important factor that determines the repair pathway of DNA double-strand breaks (DSBs). It antagonizes BRCA1 and inhibits the terminal excision of DNA double-strand breaks, thus favoring non-homologous end joining (NHEJ). Conversely, the deletion of 53BP1 has shown advantages for HR by potentially facilitating DNA DSB terminal excision and RAD51 recruitment in the absence of BRCA. In tumor samples, low expression of 53BP1 was associated with a poor response to PARPis in patients with HRD ovarian cancer. In triple-negative breast cancer models, the absence of 53BP1 compensated for BRCA defects, reduced chemosensitivity, and was associated with a poor prognosis. This indicates the clinical significance of testing the 53BP1 status in patients to assess the effectiveness of PARP inhibition. Similarly, the deletion of the downstream factor 53bp1 (rev7) also showed a recovery of HR in BRCA-deficient cells, leading to PARPi resistance. How to alleviate this resistance mechanism remains to be seen. It can be speculated that, in this case, undiscovered factors that activate or stabilize 53BP1 could be used to restore drug efficacy.
Another possibility is the loss or reduction of PARP-1 expression in tumor cells, as they inherently lack a drug target. This is particularly true for PARP capture mechanisms that rely on DNA-binding complex formation. This indicates that the stable PARP-DNA complex contributes to the cytotoxic effects of PARP inhibition. Other resistance mechanisms are associated with a dynamic balance between PARP-1 and PARG. The latter inhibits PARylation and effectively acts as a physiological PARPi, which is required to completely block PARylation in PARPi-treated cells.
In addition to the general drug efflux mechanism involving p-glycoprotein, PARPi resistance may rely on other potential "druggable" factors that compromise PARPi efficacy. Therefore, if the drug resistance mechanism can be circumvented, the sensitivity of PARPi can be restored by combined treatment.
Inhibition of cyclin-dependent kinase (CDK) is thought to make BRCA cells susceptible to PARPis. They play a key role in cell cycle progression and DNA damage control and also directly affect BRCA1/2. CDK1 promotes the mitotic process by binding to cyclin B1. Phosphorylation of BRCA1 by CDK1 is also important for the activation of downstream signaling and lesion formation. In turn, loss of inhibition of CDK1 impairs BRCA function and produces a "cancer" state. In breast cancer cells, impairment of CDK1 activity (by elimination or inhibition) has been shown to sensitize cells to PARPis in BRCA tumors. This has been shown to be an attractive drug target due to the number of CDK inhibitors on the market. Given CDK4/6 inhibitors, such as Palbociclib or Ribociclib, have been approved for hormone receptor-positive breast cancer, which may contribute to early clinical application because of the extensive clinical experience with relatively similar classes of drugs.
The other combination is the PI3K inhibitor in combination with PARPi, which is a well-studied, relatively mature approach to extending the application of PARPi. Similar to CDK1 inhibitors, PI3K inhibitors produce a "cancer" state in BRCA cells. PI3K inhibitors (BKM120) have been shown to down-regulate BRCA expression in cell lines and patient-derived xenografts. This is mediated by ERK signals through transcription factor ETS1. Another mechanism for PI3K inhibition may be impaired recruitment of DNA DSBs sites by RAD51, resulting in decreased HR. A recent Phase I clinical trial using a combination of PI3K and PARP inhibitors has shown good prospects and deserves further clinical evaluation. Given the combination of various (pre)clinical trials, there are some promising strategies to enhance the clinical application of PARPi. In particular, there is a need to improve the clinical outcomes in patients with BRCAwt tumors because PARPi have a relatively small effect in this group of patients. On the other hand, it will be crucial to more accurately predict the efficacy of PARPi in patients with HRD and, to a lesser extent, BRCA mutation carriers.
04. Future prospects
Although various treatment combinations may enhance the efficacy of PARPi and overcome resistance mechanisms, it is crucial to identify patients who are most likely to benefit. Consequently, there is a necessity to identify adequate predictive biomarkers to facilitate precise patient selection.
Screening for HRD, rather than solely depending on the clinical response to platinum-based chemotherapy, would offer more precise patient stratification. This, however, hinges on whether further characterization leads to improved treatment decisions for the patient, as well as the test's availability and reproducibility. Nonetheless, this approach could grant more patients access to PARPi therapy and the potential for additional targeted treatments.
The use of PARPi in maintenance therapy, in combination with Bevacizumab or checkpoint inhibitors, is currently being assessed in numerous ongoing clinical trials. Several trials exploring PARPi in conjunction with checkpoint inhibitors are currently available for ovarian cancer (NCT03806049, NCT03574779, NCT03598270, NCT02657889, NCT02953457, NCT03522246, and NCT03737643). It remains to be determined whether these combinations demonstrate significant clinical efficacy and improve overall survival.
References
[1] Carcinogenesis. 2012; 33(11):2026-2034.
[2] Nature. 2012; 481(7381):287-294.
[3] ESMO Open. 2018; 3(5):e000368
[4] Ann Oncol. 2017; 28(3):512-518.
[5] Lancet Oncol. 2019; 20(4):570-580.
[6] Nat Med. 2016; 22(2):194-201.
[7] Nature. 2015; 521(7553):541-544.
Statement: This article is only for sharing, if it involves copyright issues, please contact us as soon as possible, we will correct the first time, thank you!