Current location: Home > NEWS > Industry news
NEWS
PRODUCTS
Low expression of HER2 in Breast Cancer? Ultra-low expression?
News source: Release time:[2025-11-26]
Human epidermal growth factor receptor 2 (HER2) is a significant driver gene for breast cancer and an important target for targeted therapy. Currently, the expression level is primarily detected using immunohistochemistry (IHC), with in situ hybridization (FISH) technology employed to supplement the assessment of gene amplification status. This status can be categorized into two groups: HER2-positive and HER2-negative. Traditional targeted therapy against HER2 has been based mainly on HER2 positivity, which means only about 10-20% of breast cancer patients could benefit from such therapy. However, with the introduction of new antibody-drug conjugates (ADCs), the reliance on HER2 overexpression or gene amplification for traditional targeted anti-HER2 therapy has been disrupted. The conventional approach to anti-HER2 therapy has been redefined, and low expression (HER2 low) and ultra-low expression (HER2 ultralow) have been established as independent therapeutic targets for breast cancer. This development not only paves a new path for targeted therapy for traditionally "HER2-negative" patients but also broadens the population that can benefit from anti-HER2 therapy.
The concepts of HER2 low and HER2 ultralow breast cancer, both in China and internationally, primarily refer to the inclusion criteria of the Desire-Breast 04 (DB04) and Desire-Breast 06 (DB06) studies. Specifically, HER2 low is defined as an IHC score of 1+ or 2+ coupled with a FISH-negative result, while HER2 ultralow is characterized by an IHC score of 0, with no more than 10% of the infiltrating cancer cells exhibiting incomplete and weak cell membrane staining [1,2]. The ESMO Expert Consensus Statements (ECs) on the Definition, Diagnosis, and Management of HER2-low Breast Cancer, as well as the Breast Cancer Guideline 2025 issued by the China Society of Clinical Oncology (CSCO), have incorporated these concepts. This inclusion aims to better guide the interpretation of HER2 detection results and achieve more precise stratification of HER2 expression status (Fig. 1) [3, 4].
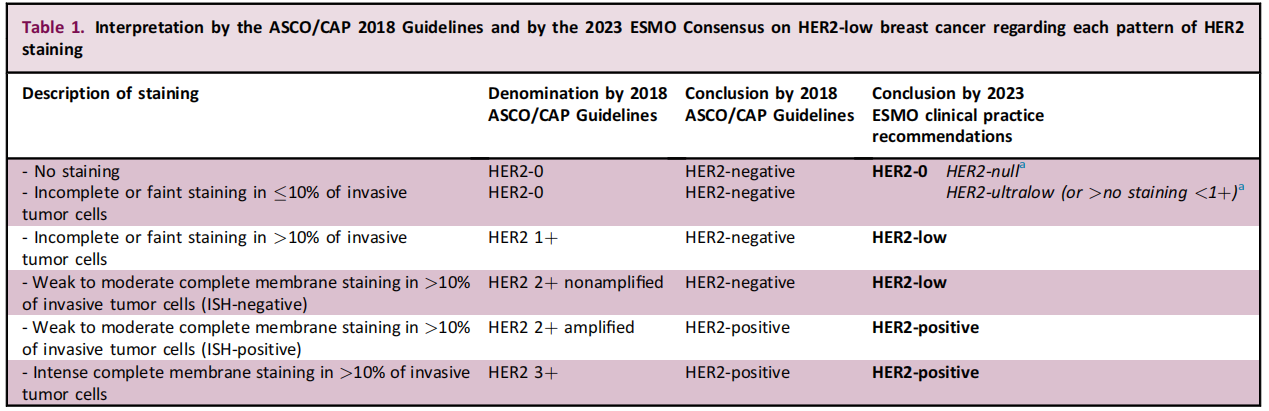
Figure 1: Definition of HER2 Low and HER2 Ultralow in the ESMO Guide
HER2 Low and HER2 Ultralow Treatment Principles
Although the HER2 expression status now includes two additional categories, HER2 low and HER2 ultralow, these are not intended to be defined as new subtypes of HER2 expression status. Currently, both domestic and international guidelines continue to use a binary classification for HER2 expression status, namely positive and negative, with HER2 low and HER2 ultralow classified as HER2 negative [3,4]. Consequently, the initial treatment regimen for patients with HER2 low and HER2 ultralow breast cancer is still recommended to be based on the molecular typing framework (Luminal A, Luminal B, HER2 positive, Basal-like), meaning hormone receptor positive (HR+) and HER2 low/ultralow patients. Following the treatment regimens for Luminal A and Luminal B types, we recommend endocrine therapy primarily with CDK4/6 inhibitors or chemotherapy primarily with Paclitaxel. After treatment failure with CDK4/6 inhibitors, ADC drugs may be considered. For patients with hormone receptor negativity (HR-), and HER2 low/ultralow, chemotherapy or chemotherapy combined with immunotherapy is recommended, drawing from Basal-like therapy protocols. Following first-line treatment failure, ADC treatment should be prioritized [4,5].
HER2 Low and HER2 Ultralow Clinical Studies
Trastuzumab Deruxtecan (T-DXd), a new generation of ADC drugs, has demonstrated potential therapeutic effects on HER2 low tumors in preclinical studies. The DESTINY-Breast 04 study, the first Phase III clinical trial to confirm the efficacy of T-DXd in the HER2 low population, enrolled 557 patients with unresectable/metastatic breast cancer in the HER2 low category who had previously undergone first-line or second-line chemotherapy. These patients were randomly assigned to either the T-DXd group or the physician's choice of chemotherapy group (TPC, such as Capecitabine, Eribulin, Gemcitabine, Paclitaxel, or albumin Paclitaxel) [1]. The results indicated that with a median follow-up of 18.4 months (data as of January 11, 2022), the median progression-free survival (mPFS) in the T-DXd group was significantly longer by 4.8 months (9.9 months vs 5.1 months, HR=0.50), and the median overall survival (mOS) was significantly longer by 6.6 months (23.4 months vs 16.8 months, HR=0.64) in the overall population compared to the TPC group (Figures 2 and 3) [1]. Based on these study results, T-DXd was approved by the US FDA in August 2022 for use in patients with unresectable or metastatic HER2 low breast cancer who have not responded to medical treatment. T-DXd was also approved by the NMPA China in July 2023 for use in adult patients with unresectable or metastatic HER2 low breast cancer who have previously received at least one systemic treatment in the metastatic disease phase, or who relapsed during adjuvant chemotherapy or within 6 months after completing adjuvant chemotherapy.
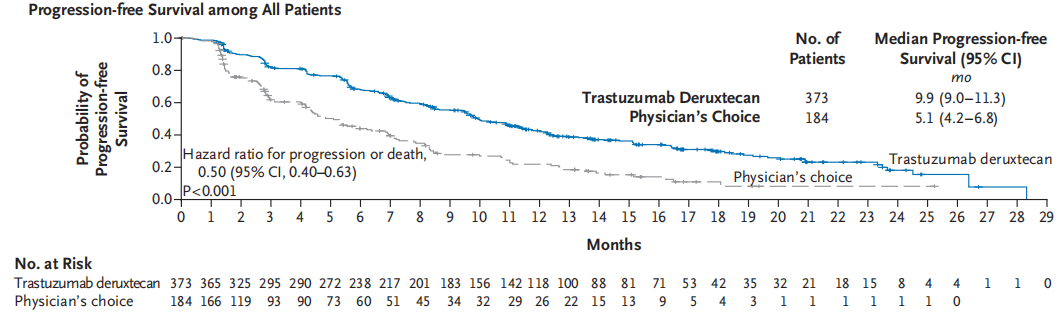
Figure 2. Destination-Break04 Study (median follow-up 18.4 months) PFS
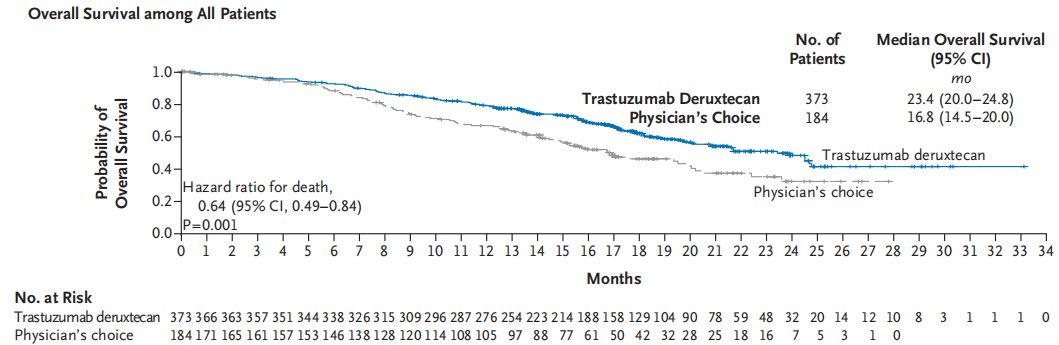
Figure 3 DESTINY-Breast 04 Study (median follow-up 18.4 months) OS
Additionally, long-term follow-up data have been published in the DB04 study, with a median follow-up of 32.0 months (as of March 1, 2023). Among the entire population, the median progression-free survival (mPFS) for the T-DXd group and the TPC group were 8.8 months and 4.2 months, respectively (hazard ratio, HR=0.36), and the median overall survival (mOS) was 22.9 months and 16.8 months, respectively (HR=0.69). These results reaffirm the superior efficacy and long-term survival benefits of T-DXd in HER2-low advanced breast cancer (refer to Figures 4–5)[6].
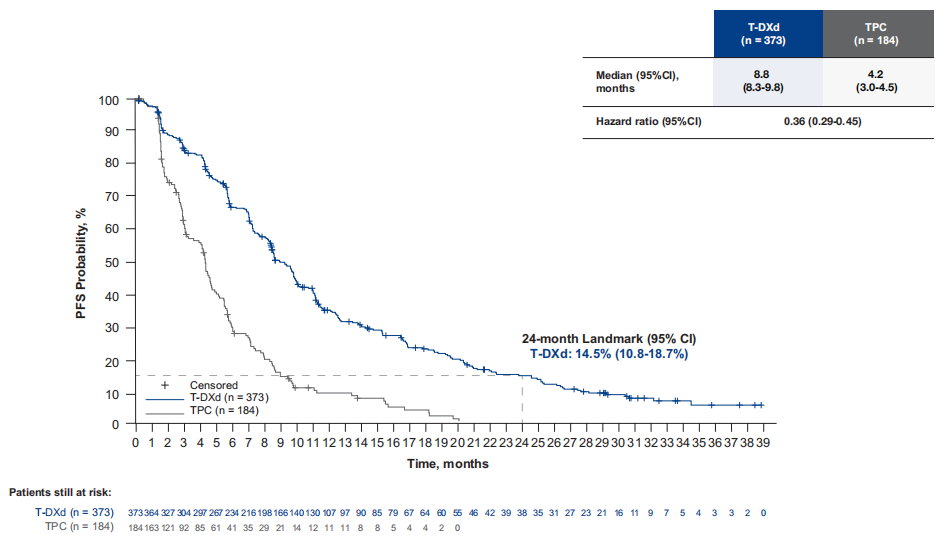
Figure 4 Destination-Break04 Study (median follow-up of 32.0 months) PFS
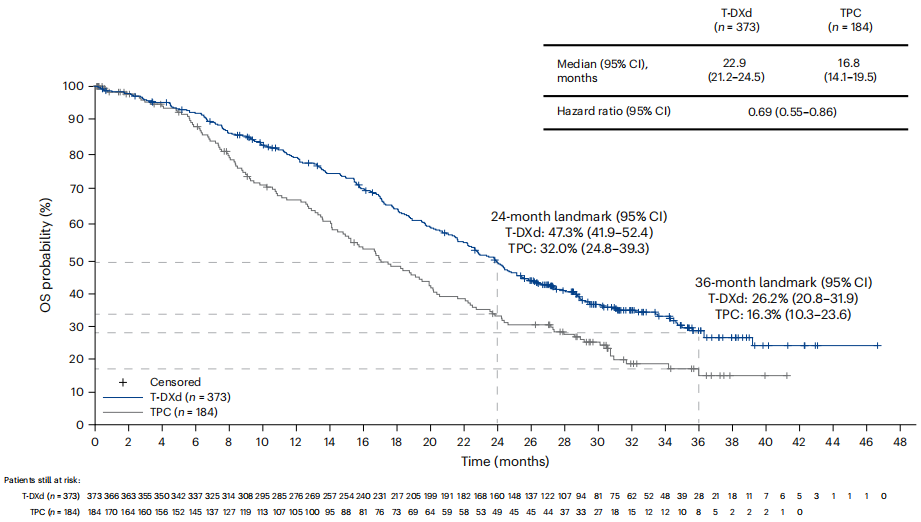
Figure 5 Destination-Break04 Study (median follow-up of 32.0 months) OS
Building on the DB04 study, DESTINY-BREAST06 expanded the HER2 treatment benefit population (HER2 ultralow) to demonstrate that T-DXd benefits the HER2 ultralow population. The DESTINY-Breast 06 study was a global, randomized, open-label Phase III clinical trial, in which 866 patients with advanced breast cancer who had previously received at least first-line endocrine therapy and had not received chemotherapy were enrolled and randomly assigned to the T-DXd group or the TPC group (such as Capecitabine, albumin-bound Paclitaxel, and paclitaxel) in a 1:1 ratio. Results show (data as of 18 March 2024) that the study has reached the primary endpoint with a significant 5.1-month (13.2 months vs 8.1 months, HR=0.62) prolongation of progression-free survival (PFS) in the T-DXd group compared with the TPC group in the HR+/HER2-low population (Figure 6). Additionally, the T-DXd group also showed a benefit trend in the HER2 ultralow population (13.2 months vs 8.3 months, HR=0.78) (Figure 7). The efficacy data from this study breaks through the dilemma of endocrine treatment for HR+/HER2-low breast cancer patients, and the benefits for the HER2 ultralow population are consistent with those of the HER2-low population, which is expected to further expand the treatment beneficiary population of Trastuzumab Deruxtecan and bring treatment hope to more patients.
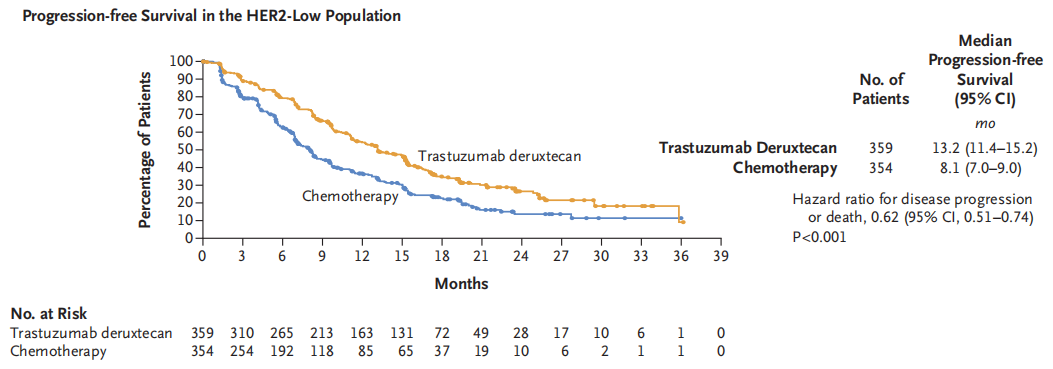
Figure 6 Destination-Break06 Study PFS in the HR+/HER2 low Population
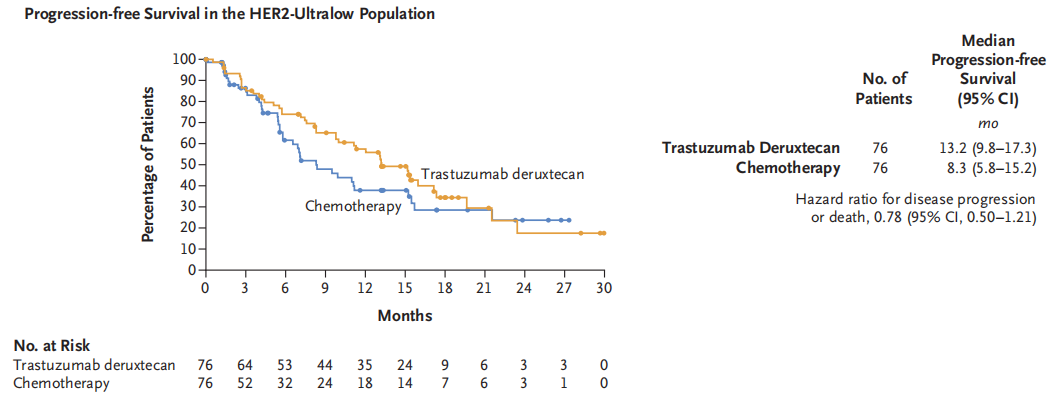
Figure 7. Destination-Break06 Study PFS in HR+/HER2 Ultra Population
04. Summary
With the breakthroughs of DESTINY-Breast 04 and DESTINY-Breast 06, the diagnostic and treatment paradigms for HER2 expression in breast cancer have been redefined. HER2 low and HER2 ultralow statuses have emerged as key targets for precise breast cancer treatment. Statistics reveal that 45%–55% of breast cancer patients exhibit a HER2 low state, suggesting that approximately half of these patients could benefit from targeted therapy against HER2. This breakthrough has shattered the previous limitation that only patients with HER2-positive breast cancer could benefit from HER2-targeted therapy, significantly broadening the population eligible for anti-HER2 treatment [5]. Nonetheless, challenges persist in the detection and diagnosis of HER2 low/ultralow statuses. Future research must continue to refine the detection methods for HER2 to more accurately identify HER2 low/ultralow breast cancer and offer patients more precise and personalized treatment plans [7].
References
[1] New England Journal of Medicine,2022,387(1): 9-20.
[2] England Journal of Medicine,2024,391(22):2110-2122.
[3] ESMO expert consensus statements (ECS) on the definition,diagnosis,and management of HER2-low breast cancer
[4] breast cancer diagnosis and treatment guidelines (2025 edition)
[5] Consensus on clinical diagnosis and treatment of breast cancer with low expression of human epidermal growth factor receptor 2 (2022 edition)
[6] Nature Medicine,2025: 1-9.
[7] Guidelines for HER2 Detection of Breast Cancer (2024 Edition)
Statement: This article is only for sharing, if it involves copyright issues, please contact us as soon as possible, we will correct the first time, thank you!