Current location: Home > NEWS > Industry news
NEWS
PRODUCTS
Revealing a "Special" Cancer: How can immunotherapy turn the tide when DNA repair "fails"?
News source: Release time:[2025-12-02]
In our cell kingdom, there is a sophisticated DNA "proofreading system" that works day and night to ensure that genetic information is copied accurately as the cells divide. This system is called DNA Mismatch Repair, MMR) system. However, when the system breaks down, errors in DNA replication keep piling up, like articles full of spelling mistakes that no one corrects. This condition is known as a mismatch repair defect (DMMR).
DMMR can cause changes in the length of short tandem repeat regions called "microsatellites" in the genome, a phenomenon known as high microsatellite instability (MSI-H). The resulting cancer is our main character today-MSI-H/DMMR tumor. Once intractable problems, they have now, by accident, become the "darling" of modern immunotherapy, staging a medical backlash.
· What are MSI-H/dMMR tumors? Understand the underlying mechanism of "DNA proofreading failure".
· Why is immunotherapy so fond of them? Uncover the biological basis of their high immunogenicity.
· Revolutionary breakthroughs in treatment: From palliative care for advanced stages to early cure, how do immune checkpoint inhibitors (ICIs) rewrite the treatment landscape?
· Future outlook: How to precisely select patients and move towards a "no-surgery" era of organ preservation.
Two main pathways contribute to the formation of MSI-H/dMMR tumors: One is the hereditary Lynch syndrome, in which patients are born with embryogenic mutations of the MMR gene; The second is acquired somatic mutations or epigenetic silencing, which are more common in sporadic cancers.
This special molecular phenotype is not uncommon, and it widely exists in a variety of solid tumors [1–7], but the prevalence difference is significant. According to the research, MSI-H/dMMR is the most common in endometrial cancer, accounting for up to 30%; This was followed by colorectal cancer (about 10%) and gastric esophageal cancer (about 9%). Interestingly, its proportion in early stage cancers is generally higher than that in advanced stage (stage IV) cancers, which may be attributed to its high immunogenicity, which enables the immune system to effectively eliminate some cancer cells with metastatic tendency in early stage.
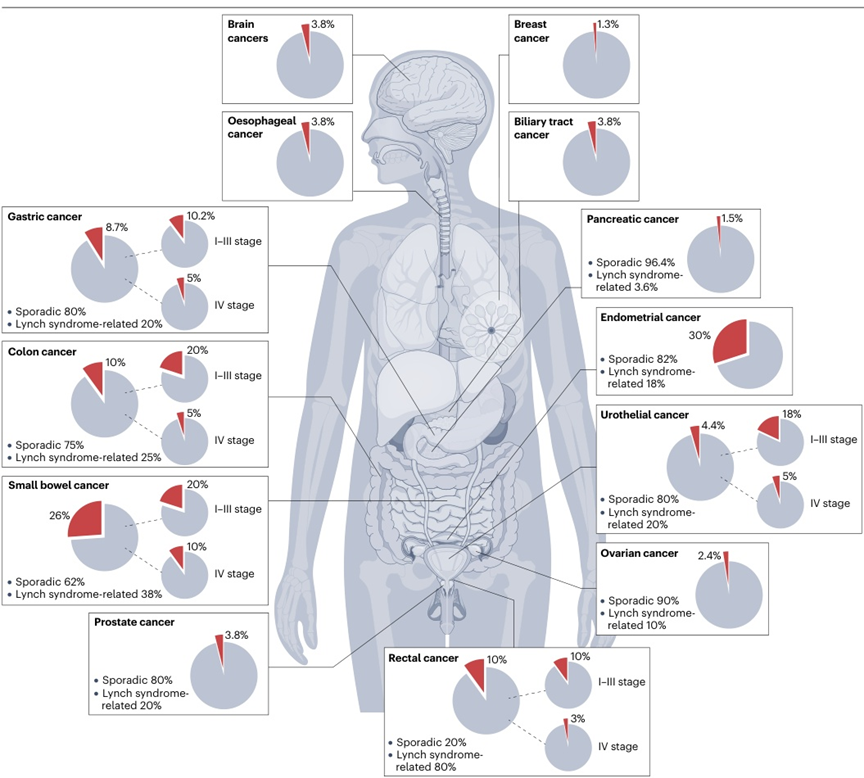
Figure 1: Distribution of MSI-H/DMMR phenotypes in different tumor types,
The red portion of the pie chart represents the ratio of MSI-H/dMMR, which is more common in gastrointestinal and gynecological tumors such as endometrial cancer, colorectal cancer, and gastric cancer.
Although the "DNA Proofreading Failure" feature of MSI-H/dMMR tumor is the root cause of cancer, it has just become its biggest "Achilles heel".
Due to defective repair function, tens of thousands of gene mutations are accumulated in these tumor cells, and the Tumor Mutational Burden, TMB) is much higher than that of common tumors. A large number of mutations, especially the frameshift mutation, will produce many normal cells do not have abnormal protein, namely "new antigen". These new antigens, like cancer cells labeled as "heterogeneous", are easily recognized and attacked by immune cells (such as T cells) in our body.
However, in order to survive, the cunning cancer cells will launch the "immune escape" mechanism, which realizes camouflage by expressing immune checkpoint proteins such as PD-L1 and sending the wrong signal of "Don't attack me" to T cells. The role of immune checkpoint inhibitors (ICIs) is to tear off this layer of "camouflage" of cancer cells, reactivate T cells, and allow the immune system to restore its strong lethality to cancer cells.
Because of this, MSI-H/dMMR tumors have the characteristics of "high mutation and high immunogenicity", making them become ideal targets of immunotherapy [8–10].
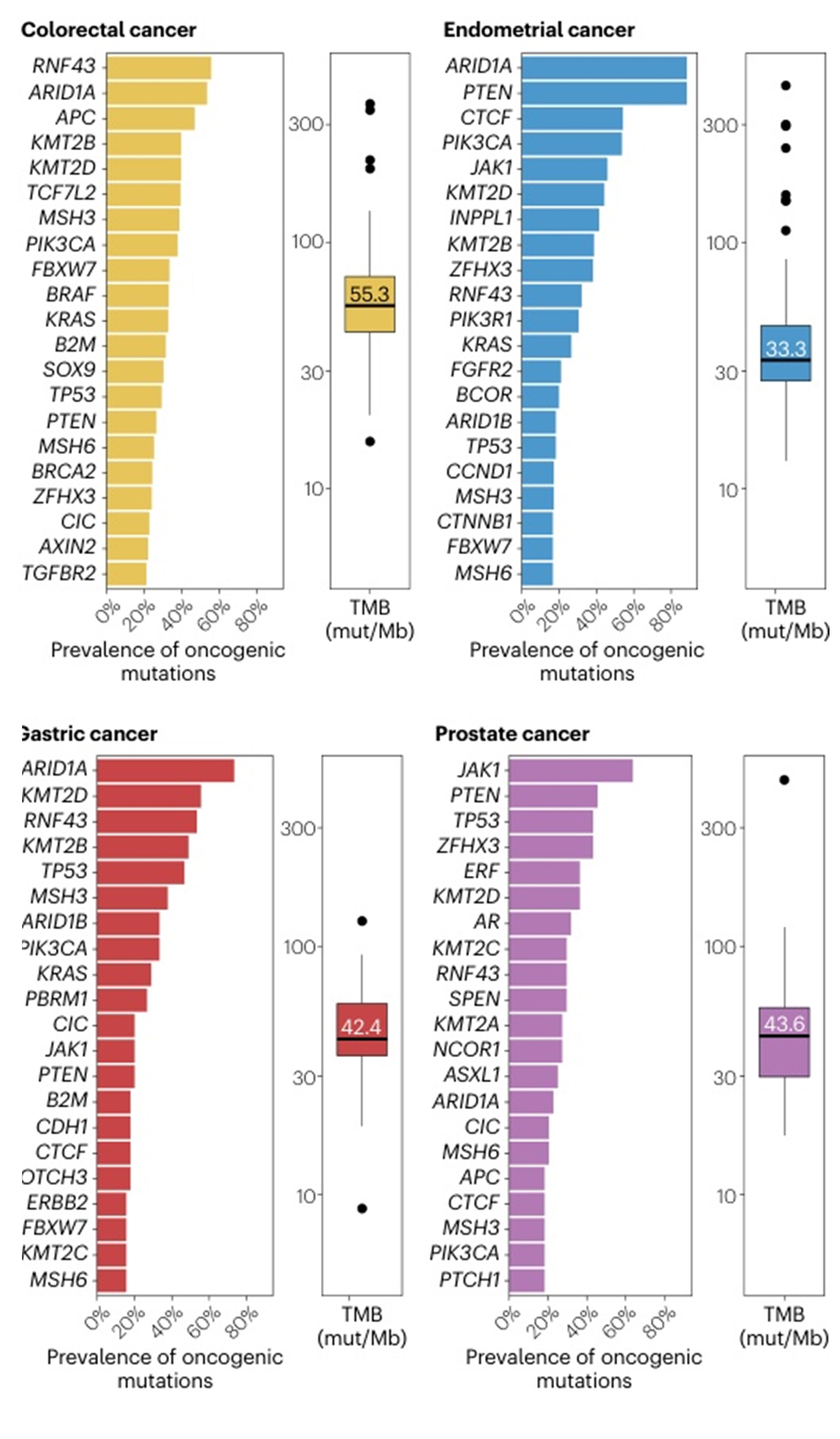
Figure 2: Most common causative mutated gene in MSI-H colorectal, endometrial, gastric and prostate cancers, with a high mutation burden common to these tumors
The appearance of ICIs has completely changed the treatment pattern of MSI-H/dMMR tumors and brought unprecedented hope to patients.
In the past, advanced MSI-H/dMMR tumors have responded poorly to conventional chemotherapy and had poor prognosis. However, a number of clinical trials (such as the KEYNOTE series of studies) have confirmed that ICIs (such as Pembrolizumab, Dostarlimab, etc.) have shown amazing efficacy in these patients, with high objective response rate and durable response. Some even advanced patients have achieved long-term survival and even "clinical cure" [11–17].
Based on this, the US Food and Drug Administration (FDA) approved the first indication of ICIs of "Histology-Agnostic" in 2017, that is, the tumor can be used as long as it has MSI-H/dMMR characteristics, regardless of which part of the body it originates from. This marks a key step in cancer treatment from an era of "organ-based" to an era of precision medicine "based on molecular markers."
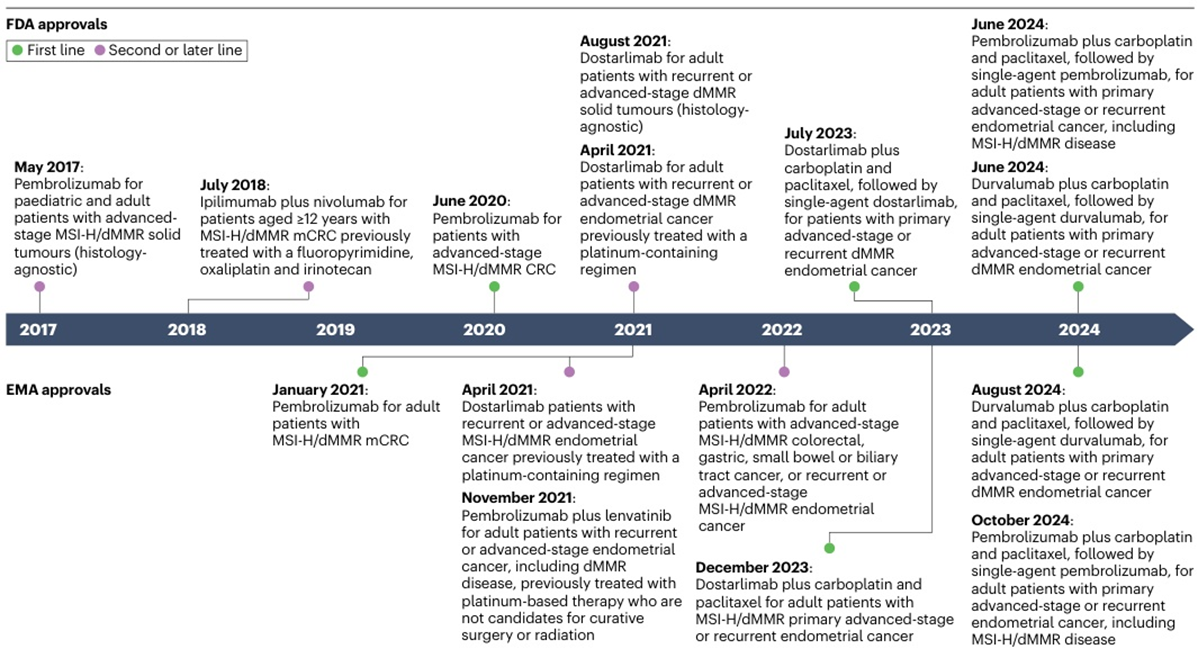
Figure 3: In recent years, FDA and EMA have approved a variety of ICIs for the treatment of MSI-H/dMMR tumors.
First-line, second-line and rear-line treatment are covered, benefiting patients with multiple cancers such as colorectal cancer and endometrial cancer.
Even more exciting is the breakthrough of ICIs in the treatment of early, resectable MSI-H/dMMR tumors. The concept of new adjuvant immunotherapy (ie, the use of ICIs before surgery) has emerged.
In the fields of colon cancer, rectal cancer and gastric cancer, multiple studies (such as NICHE-2 and NEONIPIGA) have shown that preoperative short-range immunotherapy can achieve extremely high complete pathological response rate (pCR), which means that no surviving cancer cells can be found in the surgically resected specimen. In particular, for locally advanced rectal cancer, the success of neoadjuvant immunotherapy has made "Non-operative Management" possible, and patients are expected to be cured while avoiding the pain of radical surgery and the permanent ostomy and dysfunction it brings.
Given the decisive role of MSI-H/dMMR status in prognosis and treatment choice, accurate detection is essential. At present, there are three main methods in clinical:
· Immunohistochemistry (IHC): Detects the expression of MMR proteins (MLH1, MSH2, MSH6, PMS2), which is simple to operate and low-cost, making it the preferred screening method.
· Polymerase chain reaction (PCR): Directly detects the instability of microsatellite loci, with high sensitivity and specificity.
· Next-generation sequencing (NGS): Can simultaneously assess MSI status, MMR-related genes, TMB, and other gene mutations, providing more comprehensive information.
Currently, international guidelines recommend routine MSI/MMR testing for all newly diagnosed colorectal cancer, endometrial cancer, and gastric cancer patients.
Despite the great success of ICIs, some patients are still ineffective or develop drug resistance. Future research directions include: finding more accurate prediction biomarkers to screen out the people who can benefit most; Exploring the combination strategy of ICIs with other therapies (such as targeted drugs and anti-angiogenic drugs) to overcome drug resistance; And further optimization of new adjuvant therapy in early-stage disease, allowing more patient to achieve organ preservation and high-quality long-term survival.
From a basic biological discovery to clinical practice of rewriting multiple cancer treatment guidelines, the story of MSI-H/dMMR is an excellent microcosm of the development of precision medicine and tumor immunology [18]. It tells us that an in-depth understanding of the internal mechanisms of cancer will eventually bring us a powerful weapon to overcome it.
1. Front Oncol. 2023 Oct 13; 13:1277233.
2. Ann Oncol. 2020 Oct; 31(10):1291-1305.
3. Ann Oncol. 2022 Sep; 33(9):860-877.
4. Clin Cancer Res. 2021 Mar 1; 27(5):1429-1437.
5. BMJ Oncol. 2024 Jan; 3(1):e000335.
6. N Engl J Med. 2022 Jun 23; 386(25):2363-2376.
7. Sci Rep. 2022 Nov 28; 12(1):20495.
8. Cancer Discov. 2012 May; 2(5):401-4.
9. Bioinformatics. 2014 Apr 1; 30(7):1015-6.
10. JCO Precis Oncol. 2017 Jul; 2017:PO.17.00011.
11. Food and Drug Administration. FDA grants accelerated approval to dostarlimab-gxly for dMMR advanced solid tumors (2023).
12. Food and Drug Administration. FDA grants accelerated approval to pembrolizumab for first tissue/site agnostic indication (2017).
13. J Clin Oncol. 2020 Jan 1; 38(1):11-19.
14. J Clin Oncol. 2022 Mar 1; 40(7):752-761.
15. Ann Oncol. 2022 Sep; 33(9):929-938.
16. J Clin Oncol. 2020 Jan 1; 38(1):1-10.
17. Ann Oncol. 2024 Aug; 35(8):728-738.
18. Nat Rev Clin Oncol. 2025 Jun; 22(6):385-407.