Current location: Home > NEWS > Industry news
NEWS
PRODUCTS
Did traditional standards miss 76% of high-risk patients? MRD leads the new era of precision immunity after bladder cancer surgery
News source: Release time:[2026-02-11]
There are more than 570,000 new cases of bladder cancer annually, of which approximately 25% are muscle-invasive bladder cancers (MIBC). For cisplatin-eligible patients, the standard treatment is neoadjuvant chemotherapy (NAC) followed by radical cystectomy (RC). Nevertheless, nearly half of the cases relapse within two years. Current risk assessments are unable to reliably identify the highest-risk individuals.
Currently, adjuvant immunotherapy has been demonstrated to improve the disease-free survival rate of high-risk patients [1]. Therefore, the European Medicines Agency has approved the use of adjuvant immunotherapy for high-risk PD-L1-positive patients.
The variability of pathological and clinical assessments and the limited accuracy of PD-L1 expression prediction increase the risk of overtreatment or undertreatment. This underscores the necessity for reliable biomarkers to detect minimal residual disease (MRD), guide adjuvant treatment, and enable timely intervention. Such biomarkers may also support treatment de-escalation in low-risk patients, reduce toxicity, and optimize the utilization of medical resources [2].
TOMBOLA is a Danish multicenter, open - label, single - arm phase II clinical trial evaluating circulating tumor DNA (ctDNA) assays for guiding postoperative immunotherapy. Patients with low–risk and high–risk MIBC (cT2 - 4aN0 - 1M0) treated with neoadjuvant chemotherapy (NAC) and radical cystectomy (RC) are monitored by ctDNA detection of tumor cells after surgery. ctDNA - positive (ctDNA+) patients start using Atezolizumab for up to 1 year regardless of the imaging results; ctDNA–negative (ctDNA -) patients only receive immunotherapy after X - ray - detected metastasis.
The primary endpoint is the complete response (CR) to initiate immunotherapy when ctDNA is positive, regardless of whether CT detects metastasis or not. CR is defined as the ctDNA clearance rate of the last available plasma sample during treatment, and there is no disease shown on the imaging.
Secondary endpoints include recurrence - free survival (RFS; from RC to disease recurrence or death), overall survival after RC (OS) in patients with biochemical recurrence, immunotherapy response stratified by biomarkers, clinical recurrence - free survival in patients with decreased or stable ctDNA levels during treatment, and NAC response (pathological downgrade to T0 or <T2) and its correlation with ctDNA.
There were a total of 192 patients, with 178 evaluable patients in the intended treatment population (median follow - up of 34 months). Among them, 104 (58%) had a ctDNA+ result within 2 years after RC, and 63% had it within 4 months. The median time from ctDNA detection to image confirmation of recurrence is 90 days (range from - 61 to 961 days). Among ctDNA+ patients, 84 people completed Atezolizumab treatment, and scans and ctDNA analysis were available for evaluation of the primary endpoints. 60% of patients (50 out of 84) reached the primary endpoint of complete remission. The one - year recurrence - free survival rate of ctDNA - patients was 97%, and that of ctDNA+ patients was 76%. Pre-designated biomarker analysis showed that ctDNA status and level, risk stratification, and immune-related gene expression characteristics were all related to recurrence risk and immunotherapy response. The treatment was well-tolerated, and there were no new safety risks.
For the first time, this study systematically evaluated the feasibility and effectiveness of using circulating tumor DNA (ctDNA) to guide patients with muscle - invasive bladder cancer to make immunotherapy decisions after radical cystectomy, and provided strong evidence for personalized and precise postoperative management.
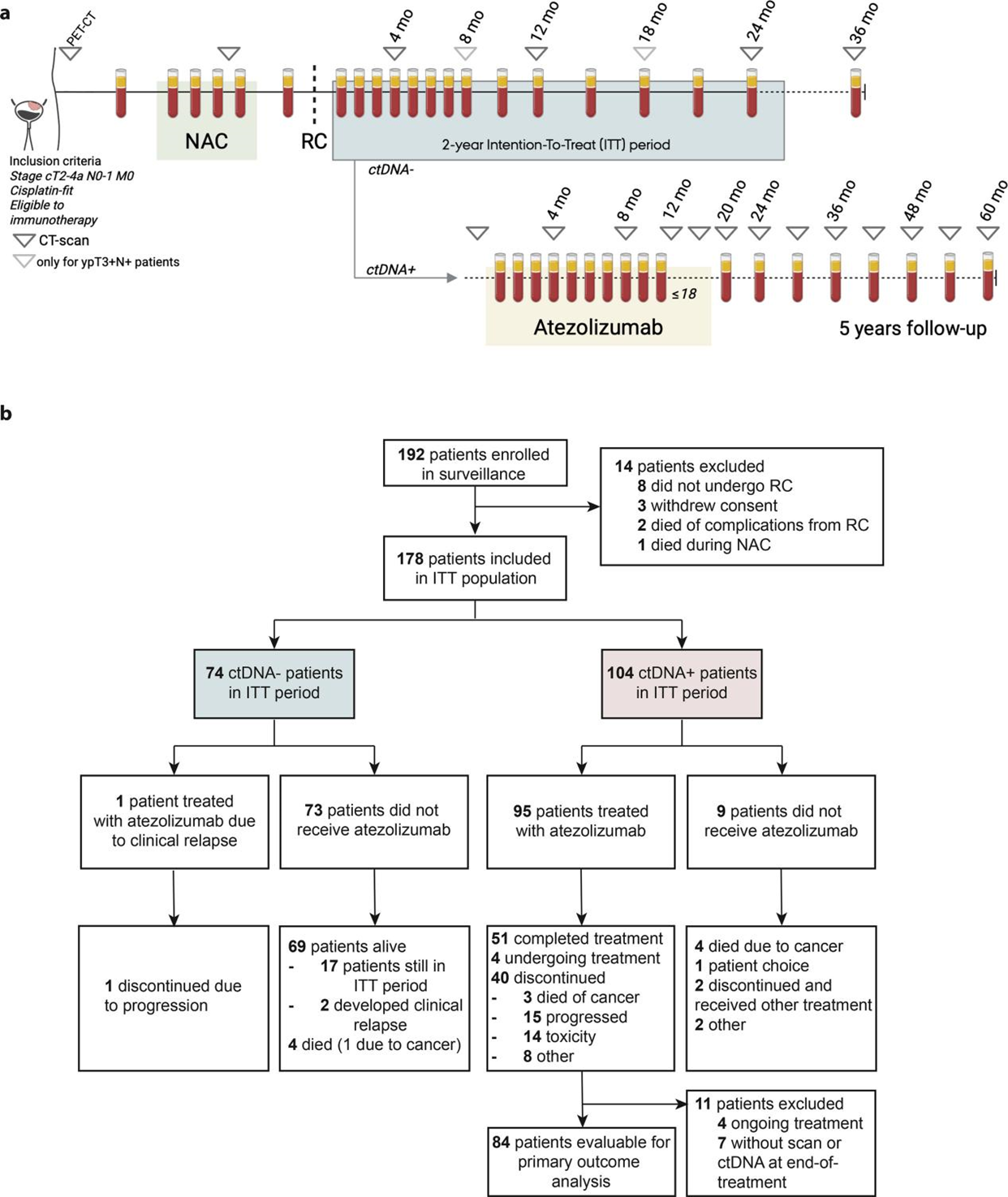
Research design, methods, and patient recruitment
In this study, the relationship between the status of tiny residual lesions (ctDNA) and the risk of recurrence was analyzed in 178 patients. The results showed that during the two - year follow - up period after the operation, a total of 58% of patients tested positive for ctDNA.
It is worth noting that among these ctDNA - positive patients, according to existing clinicopathological standards, only half of the patients are classified as traditional high - risk groups. At the same time, the proportion of high - risk and PD - L1 - positive conditions is only 24%. This shows that if one simply relies on current clinical guidelines for adjuvant immunotherapy decision - making, more than three - quarters of ctDNA - positive patients will lose the opportunity to receive intensive treatment.
On the other hand, in the patient population with persistent negative ctDNA, although 30% of patients were classified as high - risk in traditional pathological typing, there was no case of metastatic recurrence in this group of patients during the follow - up period. This result further suggests that ctDNA has an irreplaceable role in identifying the true risk of recurrence.
In terms of therapeutic intervention, after Atezolizumab was given to 84 ctDNA - positive patients with evaluable primary endpoints, 60% achieved complete remission in both molecular and imaging. The analysis shows that the dynamic changes in ctDNA levels are closely related to the recurrence and non - recurrence survival of imaging. Regardless of the low - risk or high - risk subgroups in the traditional risk stratification, the removal of ctDNA indicates a better survival outcome, which also suggests that immunotherapy can still work at very low tumor load.
At the same time, the treatment downgrade strategy has also shown good safety in ctDNA - negative patients. Follow - up data one year after the operation showed that the recurrence - free survival rate of this group of patients reached 97%, and the total survival rate reached 100%. This confirms that even if the patient meets the traditional high - risk pathological characteristics, as long as the ctDNA continues to be negative, exemption from adjuvant immunotherapy does not affect their good prognosis, thus effectively avoiding unnecessary over - treatment[3].
The TOMBOLA trial announced its first results, with a median follow - up period of 34 months. The data show that it is safe to implement a treatment downgrade strategy for ctDNA - negative patients based on the results of liquid tumor biopsy. The recurrence - free survival rate (RFS) of one year after surgery reaches 97%. The results suggest that current clinical guidelines may lead to some patients receiving over - treatment, because regardless of whether adjuvant immunotherapy is given or not, the prognosis of ctDNA - negative patients is not affected.
In ctDNA - positive patients, the RFS after one year of treatment with Atezolizumab was 75%. Regardless of the traditional risk stratification (high - risk and low - risk groups), the efficacy was observed. The ctDNA clearance rate reached 60% during treatment, which was significantly higher than the response rate reported in previous immunotherapy studies based only on imaging evaluation. This difference is most likely due to the fact that ctDNA, as a molecular marker, can reflect treatment responses earlier and more sensitively than radiological assessments.
In summary, the TOMBOLA trial has shown that ctDNA - positive patients (including traditional low - risk populations) can benefit from early immunotherapy. For ctDNA - negative patients, even if they meet traditional high - risk criteria, adjuvant therapy can be safely exempted without affecting the prognosis. This kind of biomarker - oriented individualized strategy is expected to reduce over - treatment while guaranteeing efficacy, improve the quality of life of patients, and promote the rational use of medical resources.
Statement: This article is only for sharing. If it involves copyright and other issues, please contact us as soon as possible and we will correct it as soon as possible. Thank you!