



According to GLOBOCAN data, in 2022, there will be 661,000 new cases of cervical cancer and 348,000 deaths worldwide. The number of new cases and deaths ranks first in gynecological tumors[1]. The vast majority of cervical cancer is caused by persistent infection of high-risk HPV ( hrHPV ), and the E6 and E7 genes of the virus may be integrated with the infected patients DNA. hrHPV usually includes 14 types, of which HPV 16/18 account for more than 70%[2]. In addition to cervical cancer, a certain proportion of head and neck squamous cell carcinoma, anal cancer, vaginal cancer, vulvar cancer and other cancers are caused by HPV infection.
Circulating tumor DNA ( ctDNA ) is a tumor genomic fragment released into the circulatory system by tumor tissues, cells, etc. HPV integrated into the genome of the infected person is also released in the form of ctDNA [3]. Several guidelines have recommended ctDNA for the evaluation of Molecular Residue Disease ( MRD ). ctHPV is not interfered by wild-type background in circulating cell-free DNA and is easier to detect and identify than point mutations.
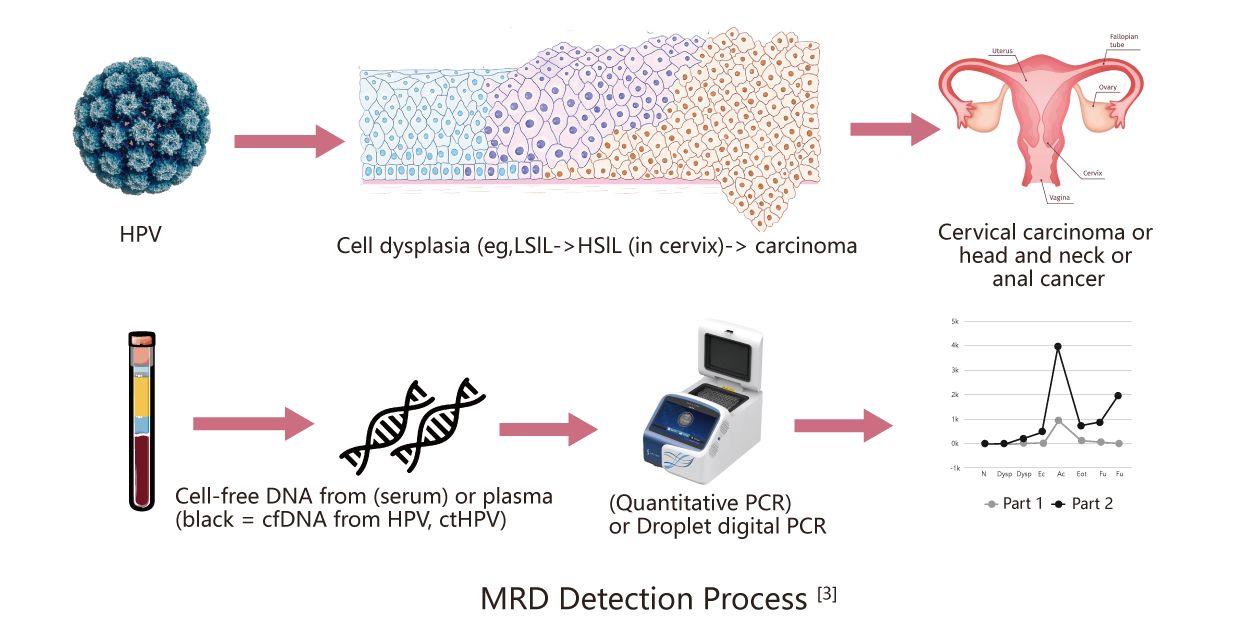
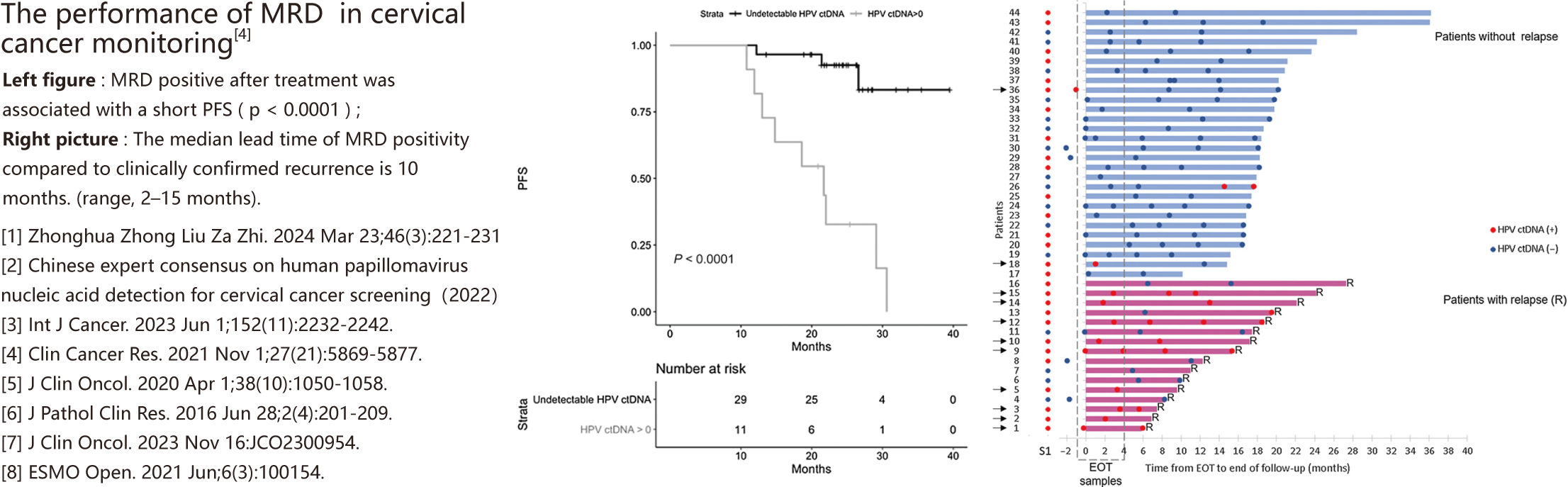
A number of studies have used MRD for the prognosis and monitoring of cervical cancer . Studies have found that patients with MRD positive at the treatment endpoint and/or during monitoring have poor survival data, and MRD can detect recurrence earlier than conventional methods [4-5]. Some other studies have suggested that the sensitivity of MRD based on digital droplet PCR ( ddPCR ) is better than that of realtime fluorescence quantitative PCR ( qPCR ) [6]. The detection performance of ddPCR is similar tohigh-throughput sequencing [7], and the method is simple and the cost is lower.
※:Research Use Only .
It is suitable for patients with cervical cancer caused by HPV 18 infection.:
(1)Auxiliary Prediction Of Therapeutic Effect : Multiple studies have confirmed that patients with MRD positive at the end of treatment and/or during monitoring have worse progression-free survival ( PFS ), disease-free survival ( DFS ), and overall survival ( OS )[4-5][8];
(2)Recurrence Monitoring After Treatment Endpoint : MRD can provide earlier indications of recurrence compared to conventional methods. For cervical cancer patients, the median lead time for detecting potential recurrence via MRD is 10 months prior to clinical confirmation [4-5].
Monitoring Scheme ( For Reference Only )




 闽公网安备35021202000745
闽公网安备35021202000745