When P53 “IHC” and “TP53” Yield the Same Result: The Clinical Points of Molecular Classification of Endometrial
The molecular classification of endometrial cancer has become a routine diagnostic item recommended by the NCCN, CSCO, and ESGO - ESTRO - ESP guidelines. Among these, the p53 abnormal subtype is a key target for precise diagnosis and treatment due to its high aggressiveness and poor prognosis. Currently, this subtype is mainly determined clinically in two ways: ① P53 immunohistochemistry (IHC) to detect protein expression; ② Detection of P53 pathogenic mutations by next - generation sequencing (NGS). However, it is not rare for the results of these two methods to be inconsistent. The initial consistency rate is only 88.6%[1]. After pathological review, there is still a disagreement rate of 5% - 6.5%, which poses challenges to subtype determination, risk stratification, and treatment decision - making.
PART 01 Disagreement Mechanism
I. Essential Biological Differences: Different Dimensions of Protein Expression and Gene State
P53 IHC reflects protein expression and localization, while TP53 gene detection directly analyzes sequence variation. The essential difference in the detection dimensions of the two is the core reason for the disagreement.
Functional Heterogeneity of Missense Mutations: TP53 missense mutations account for two - thirds of all mutation types, and 92.7% are concentrated in the DNA binding domain (DBD). Different site mutations have different effects on protein structure. For example, hotspot mutations such as R175H and R273C can lead to protein conformational changes, an extended half - life, and strong diffuse positivity in IHC. In contrast, some missense mutations (such as some VUS mutations) are pathogenic mutations, yet the protein still retains the wild - type expression pattern, resulting in the divergence of “gene mutation - protein negative” (the main type among different pathogenic cases).
Tumor Heterogeneity and Subclonal Expression: 12% of inconsistent cases are due to tumor subclonal differences. There may be both mutant and wild - type cell populations in the same lesion. IHC is prone to misjudgment as wild - type due to sampling deviation, while NGS can capture overall mutation information. A study by Zhejiang Cancer Hospital found that 9.7% of patients with TP53 missense mutations had subclonal expression patterns, indicating that the boundary between mutant and wild - type regions in the same tissue was clear[2].
Compensatory Mechanism of Tumor Cells: Tumor cells themselves have a “self - regulating” compensatory mechanism. When the binding of the p53 protein to the negative regulatory factor MDM2 is weakened, the cells will upregulate the expression of the p63 homologous protein to compensate for the functional defects of p53, so that the p53 protein can still show wild - type expression in immunohistochemistry. MDM2 expression is normal in low - invasion tumors, which can regulate the stability of p53 and avoid the accumulation of mutant proteins.
II.Interference factors at the technical level
1.Limitations of IHC detection: Antibody specificity, improper tissue fixation (formaldehyde fixation> exceeding 48 hours leads to antigen loss), tumor cell content less than 30%, etc., may result in misjudgment of the results.
2.Technical deviation of genetic testing:
Insufficient sequencing coverage: Mutations may occur in exons 1 - 11 of the TP53 gene. If the NGS panel does not cover all exons (especially exons 1, 9 - 11), rare mutations may be missed, leading to the divergence of “gene negative - protein positive”;
Impact of sequencing depth: Low - abundance mutations (mutant allele frequency VAF < 5%) require a sequencing depth of ≥500× for stable detection. If the sequencing depth is<300×, the missed detection rate can reach 35%;
Sample quality problems: DNA degradation of FFPE samples (fragment length<100bp) and tumor cell content <20% will cause the mutant signal to be diluted by normal cells, resulting in false negative results.
3. The interpretation dilemma of variation of unknown significance (VUS): TP53 VUS variation accounts for 7% - 10% of all test results. The pathogenicity of such variation is difficult to determine by sequence prediction, resulting in a decrease in the correlation between IHC and genetic test results. A 2025 PubMed study indicated that approximately 38% of TP53 VUS variants have been functionally verified as pathogenic mutations, but only 45% of them are manifested as IHC mutant patterns, and the remaining 55% are still wild - type expressions, forming a special type of “gene suspected mutation - protein negative” divergence, which causes great confusion in clinical interpretation.
PART 02 Clinical significance: the prognostic Value and classification implications of inconsistent results
I. Key reference for prognostic stratification
A study of 103 patients with TP53 missense mutations conducted by Zhejiang Cancer Hospital revealed the following:
1. The inconsistency rate between genes and proteins is as high as 41.7%
Among the 103 patients, 43 cases (41.7%) exhibited inconsistent expression of TP53 genotype and p53 IHC. There are numerous high - frequency mutation sites, such as R273C, R175H, R248Q, etc.
2. Patients in the inconsistent group were younger and had lower tumor aggressiveness
- Age: 88.9% of patients in the inconsistent group were<under 60 years old, which was significantly lower than that of the consistent group (56.0%).
- Histological classification: 72.2% of the inconsistent group were “non - invasive subtypes”, which was higher than that of the consistent group (32.0%).
- FIGO staging: The inconsistent group tended to be in the early stage (phase I–II).
3. Survival trend: The inconsistent group has a better prognosis
Although it did not reach statistical significance (P = 0.057), the inconsistent group showed a better trend in disease - free survival (DFS), suggesting that gene - protein inconsistency may be associated with better clinical outcomes.
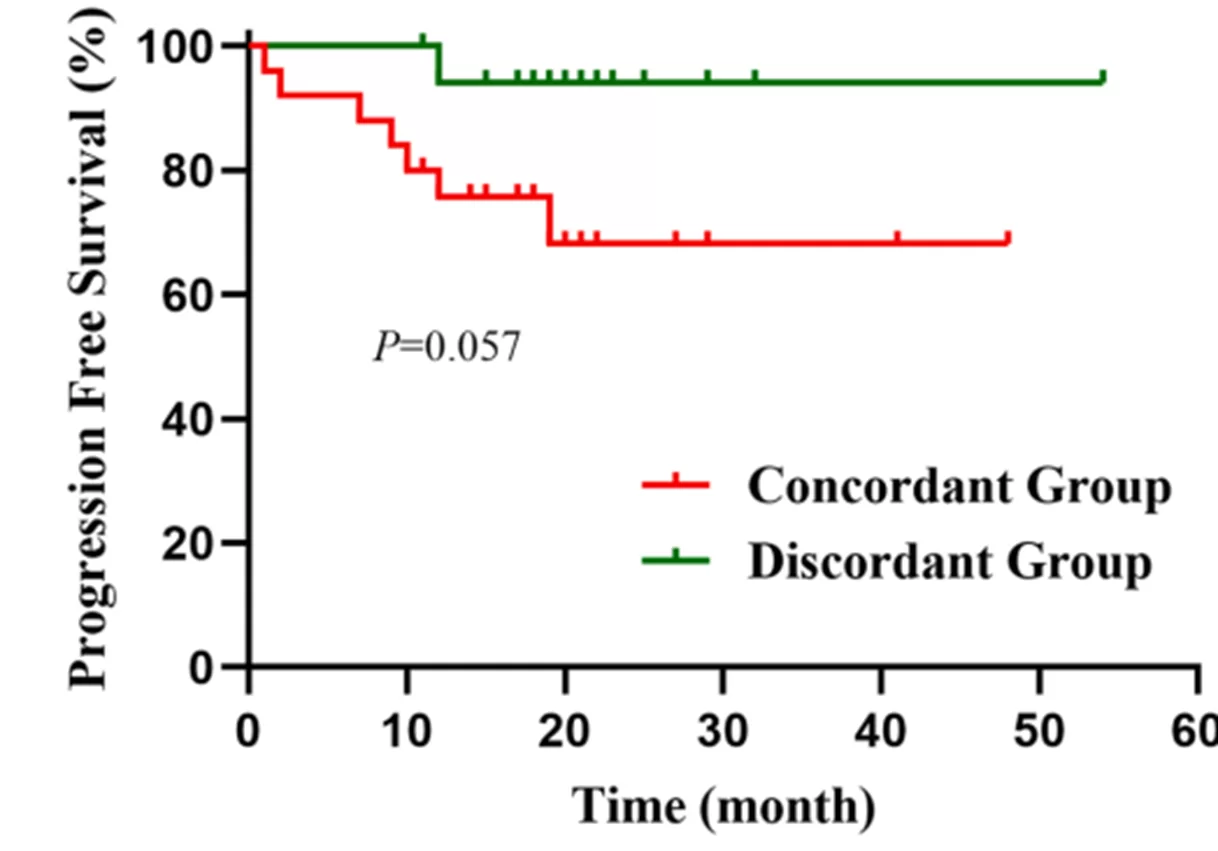
Comparison of disease - free survival of the consistent group and the inconsistent group[2]
II. The core principles of classification judgment
The molecular classification system in the guidelines' consensus is well - defined: p53 abnormal subtypes necessitate the exclusion of POLE mutations and mismatch repair defects (dMMR)/MSI, and confirmation through P53 IHC or TP53 genetic testing. Nevertheless, considering the diverse disease cases, the latest research recommends:
1.For patients with a "gene - mutated, protein - wild - type" status and no other high - risk factors (low - grade, early - stage, and no vascular involvement), a comprehensive assessment is required to determine whether to intensify treatment to avert over - treatment.
2.For cases of "gene - negative, protein - mutant", the IHC slides need to be re - examined to rule out technical errors. If the mutant expression pattern is verified, it should still be classified as a p53 abnormal subtype.
PART 03 Summary
Inconsistent results between P53 immunohistochemistry (IHC) and TP53 genetic testing are a prevalent issue in the molecular classification of endometrial cancer. The fundamental reason lies in the inherent biological differences (different dimensions of protein expression and gene status) and interference factors at the technical level. The molecular classification of endometrial cancer has been gradually refined. Molecular classification has not supplanted morphology and immunohistochemistry but has instead formed an integrated diagnostic model of "molecular characteristics + morphology + immune markers". Clinical practice has verified that the integration of these three elements can significantly enhance the accuracy of diagnosis and the value of treatment guidance, while also avoiding misjudgments caused by a single indicator. Therefore, for the classification of endometrial cancer, the model of "technical standardization + comprehensive result interpretation + multi - disciplinary team (MDT) decision - making" is employed: technical errors are minimized by standardizing the testing process, subtypes are determined based on population characteristics and clinical high - risk factors, and new drugs are utilized to offer personalized treatment options for different cases of the disease. In the future, with the optimization of detection technology and breakthroughs in treatment targets, it is anticipated to further narrow the detection gap and achieve precise stratification and personalized treatment in the true sense.
References
[1] Machuca-Aguado J, Catherwood M, Gonzalez D, McCluggage WG. Correlation between P53 immunohistochemical staining and TP53 molecular testing in endometrial carcinomas: a detailed assessment of discrepant cases with implications for patient management. Histopathology. 2026 Feb;88(3):591-604. doi: 10.1111/his. 70029. Epub 2025 Oct 31. PMID: 41171033.
[2] Yang Y, Sun S, Xi Y, Xu C, Zhu R, Yin W, Zhan Y, Su D. Correlation analysis of TP53 missense mutations and p53 protein expression patterns in endometrial carcinoma. Hum Pathol. 2026 Jan 23;171:106056. doi: 10.1016/j. humpath. 2026.106056. Epub ahead of print. PMID: 41581707.
Statement: This article is only for sharing. If it involves copyright and other issues, please contact us as soon as possible and we will correct it as soon as possible. Thank you!




 闽公网安备35021202000745
闽公网安备35021202000745