Without Driver Alterations, Whither The Treatment Paradigm?
The treatment strategy for stage IV non - small cell lung cancer (NSCLC) lacking specific driver gene mutations is undergoing continuous and profound evolution. The 2026.3.0 version of the dynamic guidelines issued by the American Society of Clinical Oncology (ASCO) incorporates the latest clinical trial evidence as of October 2025 and offers key updates for clinical decision - making in this complex domain [1]. In particular, it focuses on the stratification of first - line treatment, innovation of back - line treatment, and advancement in the exploration of future treatment models.
First - line treatment: Precise stratification and strategic trade - off based on PD - L1 expression
The new edition of the guidelines further solidifies the status of PD - L1 expression as a core biomarker for first - line treatment decision - making. The selection of treatment strategy is no longer a mere enumeration of options but is based on the comprehensive trade - off of efficacy, toxicity, and patient characteristics at different PD - L1 expression levels.
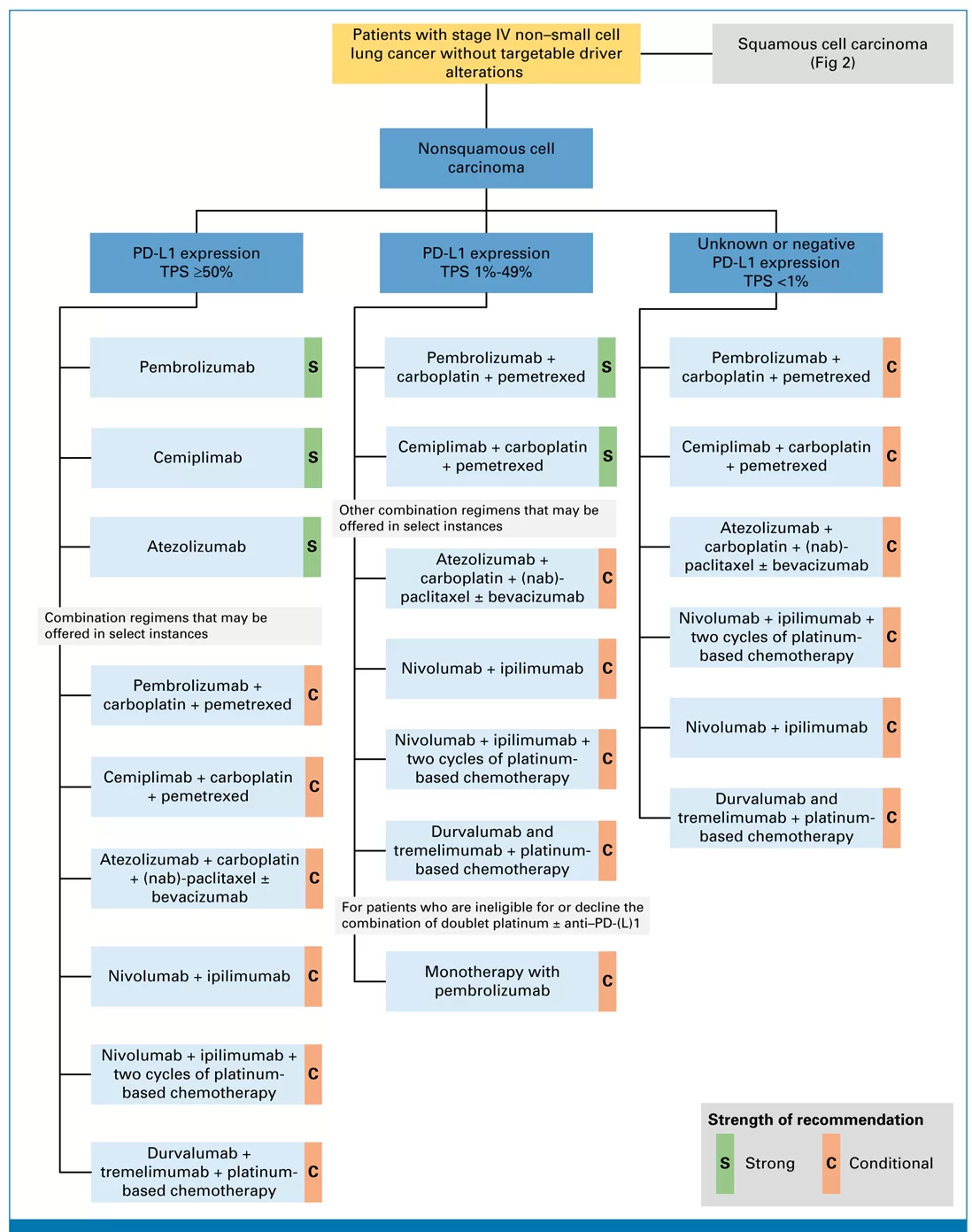
First-line treatment options for patients with stage IV non–small cell lung cancer without driver alterations and nonsquamous cell carcinoma.
For recommendations with multiple treatment options of the same evidence quality
and strength of recommendation, the decision of which agent to offer should be tailored based on discussion of efficacy and toxicity with each patient. nab, nanoparticle albumin-bound; NSCLC, non–small cell lung cancer; PS, performance status; TPS, tumor proportion score.
1. PD - L1 TPS ≥50%: Reconfirmation of the cornerstone status of immunotherapy monotherapy
For individuals with high PD - L1 expression, the guidelines reaffirm the cornerstone status of PD - (L)1 inhibitor monotherapy (Pembrolizumab, Cemiplimab, or Atezolizumab) at the level of “strong recommendation”. The confidence in this recommendation is derived from convincing long - term survival data from a number of Phase III clinical trials. For instance, the 5 - year follow - up of the KEYNOTE - 024 study indicated that the overall survival (OS) rate of Pembrolizumab monotherapy reached 31.9%[2]; similarly, the 5 - year OS rate of Cemiplimab in the EMPOWERMENT - Lung 1 study also reached 29.0%[3]. These data set a benchmark for the efficacy of immunotherapy monotherapy in this population.
It is noteworthy that the guidelines do not prioritize combined chemotherapy or dual - free regimens for this subgroup. The clinical consideration underlying this is: although combination regimens (such as KEYNOTE - 189/407) also demonstrate OS benefits in PD - L1 high - expression subgroups, there is currently a lack of direct evidence indicating their superiority over monotherapy. More importantly, combination therapy is certain to increase the risk of toxicity. Therefore, in the absence of clear evidence of enhanced efficacy, it is an important clinical principle to avoid unnecessary treatment burden. Only for specific patients with high tumor load, severe symptoms, or risk of visceral crisis, clinicians may consider combination chemotherapy to pursue a higher objective remission rate and more rapid tumor regression.
2. PD-L1 TPS 1% - 49%: The standard paradigm for immunotherapy and chemotherapy
For patients with low PD-L1 expression, the model of immune checkpoint inhibitors combined with platinum-based two-drug chemotherapy has become the indisputable standard treatment. The 5-year data of the KEYNOTE - 189 STUDY indicated that in the PD-L1 1 - 49% subgroup of non-squamous cell carcinoma, the 5-year survival rate of the Pembrolizumab combined chemotherapy group reached as high as 19.8%, whereas that of the simple chemotherapy group was merely 7.7% [4]. Similar conclusions were also confirmed in the EMPOWER - Lung 3 study of Cemiplimab. Cemiplimab combined with chemotherapy significantly enhanced survival (HR = 0.50), further strengthening the evidence-based medical foundation of the combined strategy.

3. PD - L1 TPS < 1%: Exploration and Trade - off of Diversified Combination Strategies
The treatment of PD - L1 negative patients is currently the most challenging and exploratory area. The guideline offers a variety of “conditional recommendations”, which reflect the trade - off between the efficacy and toxicity of different strategies.
Immunotherapy + Standard Chemotherapy: As a fundamental option, its effectiveness has been verified in the PD - L1 negative subgroup studied by KEYNOTE - 189 and other research. Although the 5 - year survival rate (9.6% vs. 5.3%) has been improved, the absolute benefit is restricted.
Dual Immunotherapy + Limited Chemotherapy: The CheckMate 9LA regimen (Nivolumab + Ipilimumab + 2 cycles of chemotherapy) has provided significant inspiration for this subgroup. Its design concept is to rapidly control tumors through short - term chemotherapy while leveraging the synergy of dual immunotherapy to stimulate a sustained immune response. The final analysis indicated that in the PD - L1 negative population, the 6 - year survival rate of the regimen reached 20%, far exceeding the 7% of chemotherapy alone, and its “long - tail effect” is particularly notable [5].
Four - drug Combination Regimen: The POSEIDON study investigated the model of Durvalumab + Tremelimumab + chemotherapy and also observed a trend of survival benefit, offering another option for clinical practice.
Clinical decision - making necessitates a meticulous trade - off among the patient's physical condition, comorbidities, and tolerance to different toxicity profiles.
Back-line treatment: Antibody-coupled drugs (ADCs) open a new dimension of targeting
For patients who have experienced disease progression following platinum-containing chemotherapy and immunotherapy, the efficacy of the traditional Docetaxel (±Ramucirumab) regimen is limited. A significant alteration in the 2026 edition of the guidelines is the inclusion of two ADC drugs in the recommendations, indicating that even in non-small cell lung cancer (NSCLC) without “classical” driver gene mutations, back-line treatments are commencing to transition towards the era of precise targeting based on protein expression.
New recommendation for back - line treatment (conditional): · Telisotuzumab vedotin (Teliso - V): Intended for patients with high expression of c-MET protein (defined as having 3+ intensity in ≥25% of tumor cells). The LUMINOSITY trial indicated that the objective response rate (ORR) of this patient population was 34.6%, and the median overall survival (OS) was 14.6 months, offering valuable treatment alternatives [6]. · Trastuzumab deruxtecan (T - Dxd): Applicable to patients with HER2 overexpression (IHC 3+). According to the data from the DESTINY - Lung01/03 study, at a dosage of 5.4 mg/kg, the ORR can range from 34.1% to 44.4%, and the median OS is between 11.2 and 17.1 months, demonstrating distinct clinical efficacy [7]. |
The clinical significance of this update lies in the fact that it mandates clinicians to reevaluate tissue biopsy or make use of existing samples for immunohistochemistry (IHC) testing of c - MET and HER2 when the patient's back - line treatment proves unsuccessful, with the aim of identifying new treatment opportunities for patients.
Future Direction: Evolution of Treatment Mode and Exploration of New Mechanisms
The "Future Research" section of the guide actively identifies two directions that will have a profound impact on clinical practice.
1. Subcutaneous Injection (SC) Dosage Form: Optimize Treatment Experience and Medical Resource Allocation
The subcutaneous injection dosage forms of Pembrolizumab and Atezolizumab have shown in Phase III studies (3475A - D77 and IMscin001) that they are not inferior to intravenous dosage forms in terms of pharmacokinetics, efficacy, and safety. Their core advantage lies in significantly reducing the patient's in - hospital treatment time (the "seat time" is reduced by nearly 50%)[8]. This not only enhances the patient's treatment experience but also offers a viable path for optimizing the allocation of medical resources and improving the operational efficiency of the medical system.
2. VEGF/PD - 1 Bispecific Antibody: Challenging Existing Standards
The guide gave special attention to the HARMONi - 2 research carried out in China. In this study, Ivonescimab, a VEGF/PD - 1 bispecific antibody, had significantly better progression - free survival (PFS) than Pembrolizumab in the first - line treatment of programmed death - ligand 1 (PD - L1) - positive non - small cell lung cancer (NSCLC), and a trend of overall survival (OS) benefit was observed[9]. This result implies that the synergistic blocking of the VEGF and PD - 1 pathways may bring clinical benefits beyond single - target inhibition, presenting a potential challenge to existing first - line treatment standards. The results of its global multicenter research are highly anticipated.
Clinical Inspiration
The core of the 2026.3.0 version of the American Society of Clinical Oncology (ASCO) dynamic guidelines is the advancement from "stratified therapy" to "precise stratified therapy". At the front line, the quantitative evaluation of PD - L1 expression guides the selection and trade - off among immunotherapy monotherapy, immunotherapy combined with chemotherapy, or more complex combination models. At the frontline, protein expression levels such as c - MET and human epidermal growth factor receptor 2 (HER2) are emerging as new biomarkers, facilitating the introduction of antibody - drug conjugate (ADC) drugs into clinical practice. Additionally, the guidelines once again stress the importance of integrating interdisciplinary palliative care (including innovative telemedicine models) in the early stages of the disease, which is of immeasurable value for comprehensively improving the quality of life of patients and enhancing overall survival.
References
1. J Clin Oncol. 2026 Mar;44(7):e56-e88.
2. J Clin Oncol. 2021;39:2339-2349.
3. J Thorac Oncol. 2025;20:941-954.
4. J Clin Oncol. 2023;41:1992-1998.
5. ESMO Open. 2025;10:105123.
6. J Clin Oncol. 2024;42:3000-3011.
7. Lancet Oncol. 2024;25:439-454.
8. Ann Oncol. 2025;36:775-785.
9. Lancet. 2025;405:839-849.
Statement: This article is only for sharing. If it involves copyright and other issues, please contact us as soon as possible and we will correct it as soon as possible. Thank you!




 闽公网安备35021202000745
闽公网安备35021202000745