A New Perspective on the Diagnosis and Treatment of Glioma: How Do MET Gene Changes Affect the Survival of Patients with
Introduction
Gliomas are the most prevalent malignant brain tumors, accounting for approximately 80% of all malignant brain tumors. They can be regarded as “aberrant invaders,” characterized not only by rapid growth but also by the potential variability in tumor characteristics among individual patients, which poses significant challenges to treatment. In 2021, the World Health Organization released the latest brain tumor classification standards, according equal importance to genetic test results and tissue morphology under the microscope. This change has enabled the identification of numerous concealed “adversaries,” including the focus of this article: MET genetic abnormality.
PART 1
What is MET Gene Abnormality?
The MET gene can be likened to a “keyhole” on the cell surface. Under normal conditions, cell growth signals are activated only after the insertion of a specific “key” (HGF protein). However, in certain glioma patients, there are two “variants” of the MET gene:
Fusion
The MET gene undergoes splicing with an adjacent gene (such as PTPRZ1) to form a “hybrid” gene, which produces a protein that is perpetually “active,” continuously stimulating tumor growth.
Splicing Variation
The MET gene skips exon 14 during transcription, resulting in the produced protein lacking a “regulatory” structure and being unable to undergo normal degradation, which also leads to excessive signal activation [1].
These two anomalies are collectively referred to as MET F/SVs. Among them, a particular fusion, PTPRZ1 - MET fusions (abbreviated as ZM fusions), is the most prevalent and perilous in gliomas.
Scientists collected data from 1,041 glioma patients from two large - scale databases (Chinese Glioma Genome Atlas and a targeted sequencing study), re - classified them according to the 2021 latest standards, and analyzed the MET gene status, pathological characteristics, treatment status, and survival time of these patients. The tumor tissues of all patients underwent rigorous pathological review, and key molecular markers were determined through second - generation sequencing [2].
PART 2
Which Patients Are Prone to Carrying MET Gene Abnormalities?
Among the 1041 patients, a total of 49 (4.70%) carried MET F/SVs. Among these, fusions accounted for 43.8%, and splicing variations accounted for 56.2%. ZM fusion is the most common type of fusion, accounting for 75% of all fusion events.
Interestingly, MET F/SVs are only observed in two types of gliomas: IDH mutant astrocytoma and IDH wild - type glioblastoma. Oligodendrocytoma and childhood diffuse glioma were not detected at all.
Specifically:
l In astrocytomas, the proportion of MET fusion was 3.66%, and ZM fusion accounted for 2.88%.
l In glioblastoma, the proportion of MET fusion was 2.99%, and ZM fusion accounted for 2.74%.
If astrocytomas are classified by grade, the proportion of MET F/SVs in patients with the highest grade 4 has surged to 9.18%, and ZM fusion has also reached 4.72%. More importantly, the proportion of patients with recurrent astrocytoma carrying MET F/SVs was significantly higher than that of patients undergoing initial surgery (8.5% vs. 3.2%). In relapsed grade 4 astrocytoma, the proportion of ZM fusion is as high as 5.97%—this represents the highest known risk population.
Other clinical features (age, gender, degree of surgical resection, radiotherapy and chemotherapy, etc.) did not exhibit significant differences between patients carrying and not carrying MET F/SVs, indicating that this genetic change is an “adverse signal” independent of conventional factors.
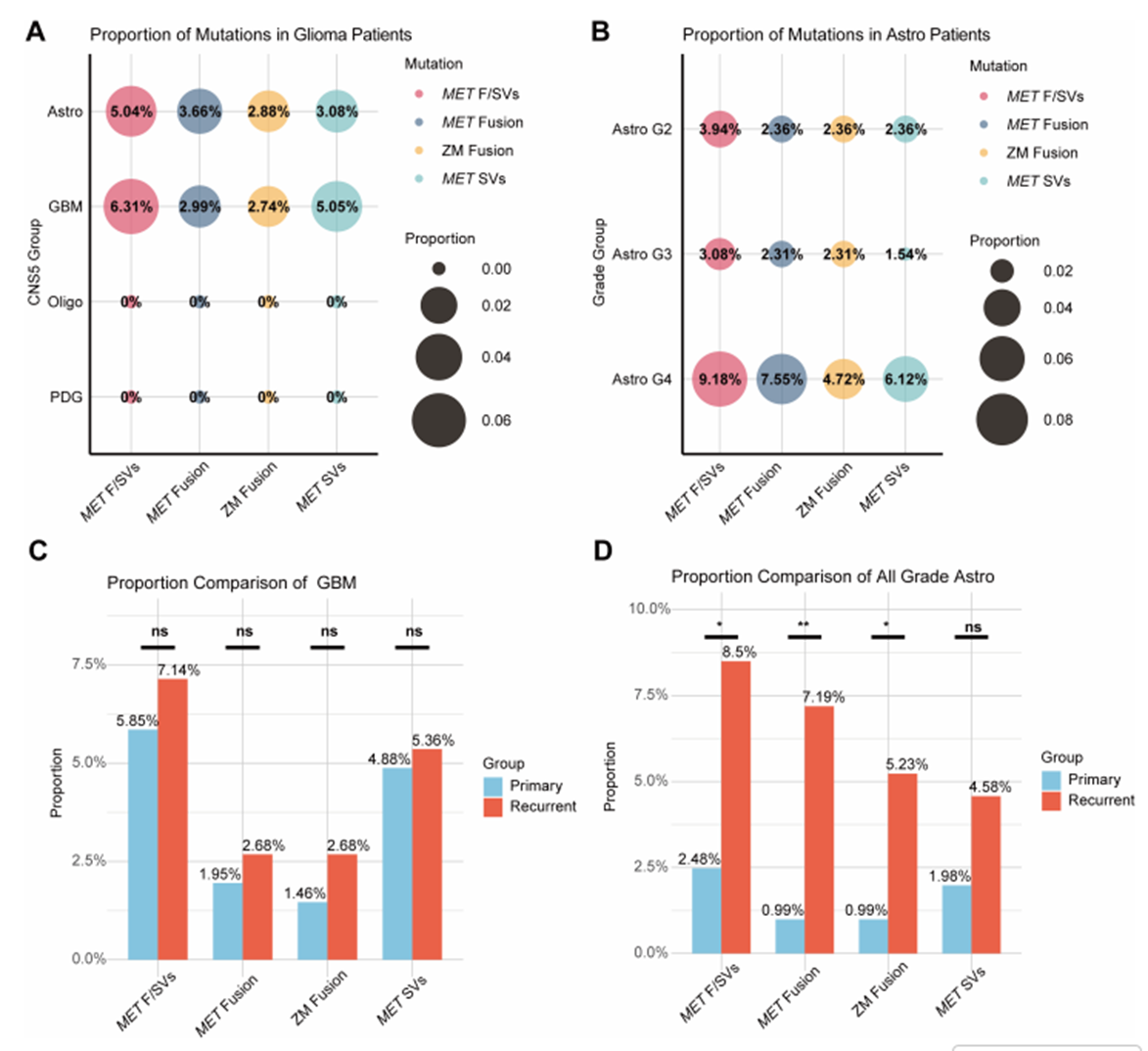
Distribution of MET gene abnormalities in different glioma types[3]
PART 3
Can abnormalities in the MET gene lead to protein overexpression?
The answer is affirmative. The researchers employed immunohistochemical staining to directly observe the MET protein in the tumor tissue. For tumors harboring ZM fusion, the MET protein exhibited strong positivity (dark color and wide distribution), whereas in those without it, the protein was scarcely detectable. RNA sequencing also confirmed that regardless of whether it is astrocytoma or glioblastoma, as long as it carries ZM fusion or MET F/SVs, the transcription level of the MET gene is significantly elevated. In other words, this genetic abnormality directly resulted in a substantial increase in the number of “keyholes,” and the growth signals received by tumor cells became increasingly stronger.
PART 4
What is the relationship between MET gene abnormality and patient survival?
First, consider the level of MET protein expression. The overall survival period and the progression - free survival period of patients with high MET expression are significantly shortened. The median overall survival of the high - expression group was only 484 days, while that of the low - expression group could reach 1430 days. In high - grade astrocytomas, this disparity is even more pronounced.
Next, examine the genetic level. Patients carrying MET F/SVs have a median overall survival of 327 days, while those without it have a median overall survival of 914 days. Patients with ZM fusion had a worse survival outcome: the median overall survival was only 250.5 days, compared with 848 days for non - carriers.
Among patients with astrocytoma, the median overall survival period (378 days) of MET F/SVs - positive patients is even shorter than that of conventional grade 4 patients (917 days). Multivariate analysis (after correcting for factors such as age, tumor classification, primary/recurrence, etc.) confirmed that MET F/SVs is an independent “death risk factor,” which increases the overall survival risk by 141% and the non - progressive survival risk by 202%.
In glioblastoma, MET F/SVs also demonstrated a tendency to shorten survival, but statistically, it did not reach a significant difference. This may be attributed to the extremely high degree of malignancy of glioblastoma itself, the generally short survival period of patients, and the small number of patients carrying the abnormality, resulting in insufficient statistical power.
PART 5
ZM fusion: The molecular subtypes that demand the highest level of vigilance
Further analysis revealed that ZM fusion is the primary “culprit” contributing to poor prognosis. In astrocytoma, the median overall survival of ZM fusion - positive patients is only 382.5 days, while that of high - grade negative patients is 855 days, and that of low - grade negative patients is up to 2861.5 days. Multivariate analysis indicated that ZM fusion increases the risk of death by more than 4 times.
In contrast, simple variable splicing (without fusion) did not show independent prognostic significance in multivariate analysis. This prompts clinicians: if ZM fusion is detected, regardless of the tumor grade, the patient should be regarded as a high - risk case, and a more aggressive treatment plan should be considered.
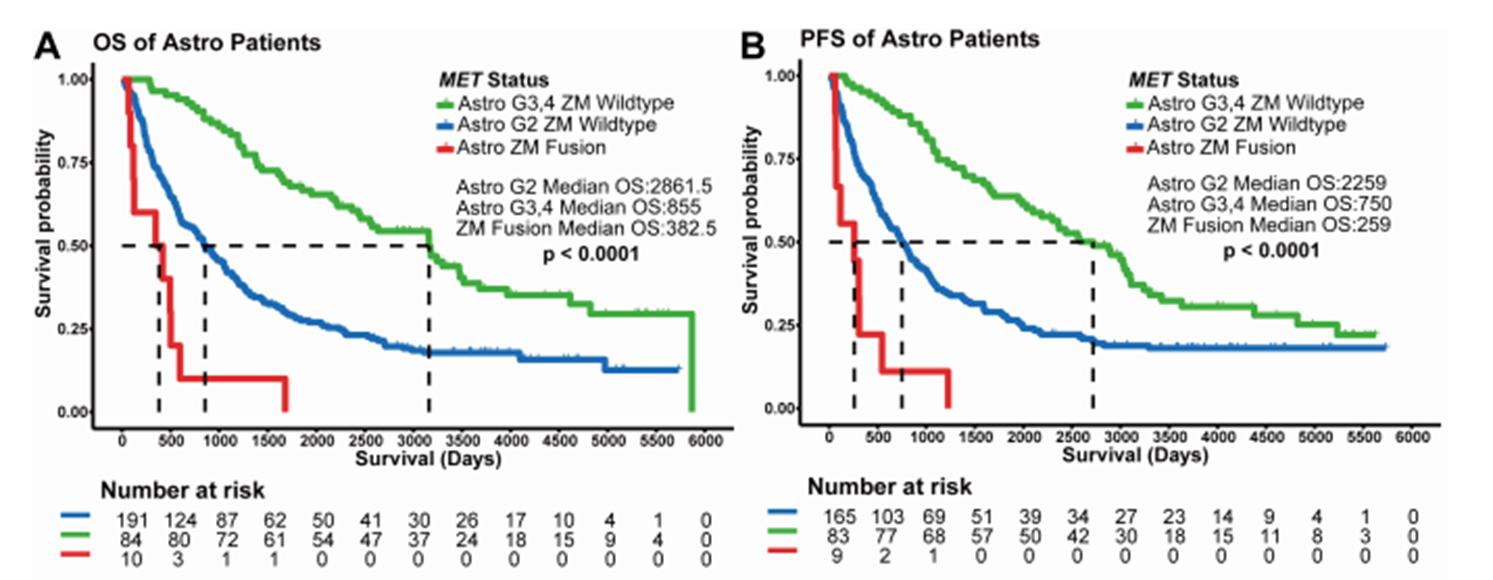
Survival curve of patients with ZM fusion in astrocytoma[4]
PART 6
What are the implications for patients?
1. Detection is of utmost importance. For patients with glioma, particularly those with astrocytoma and recurrent cases, it is recommended to conduct MET gene testing (including fusion and splicing variants) during surgery. At present, targeted drugs (a MET inhibitor) for ZM fusion have entered the clinical trial stage, and some have been approved for use. Only through testing can we determine whether this type of targeted therapy is suitable.
2. Prognostic judgment is more accurate. If MET F/SVs, especially ZM fusion, is detected, it indicates that the biological behavior of the tumor is more “dangerous.” Even if the pathological grade is not high, follow - up and treatment should be carried out according to the highest risk level.
3. The future is promising. In addition to ZM fusion, other types of MET abnormalities (such as MET amplification and MET14 exon skipping) also have corresponding targeted drugs under development. With the advancement of precision medicine, an increasing number of glioma patients will benefit from “personalized treatment guided by genetic testing.”
PART 7
Research limitations
There are also certain limitations in this study. All patients are of Asian ethnicity, which may introduce racial bias. Some early cases cannot be accurately graded due to incomplete data. ZM fusion is relatively rare, resulting in a small sample size of glioblastoma subgroups. Larger - scale, multi - center, and multi - ethnic studies are required in the future to validate these findings.
PART 8
Summary
This multicenter study involving 1,041 patients, based on the latest glioma classification criteria, systematically depicted a “panorama” of MET gene fusion/splicing abnormalities for the first time.
MET F/SVs mainly occur in astrocytomas and glioblastomas, and have the highest proportion (nearly 10%) in recurrent high - grade astrocytomas.
ZM fusion is the most significant type and the most perilous adverse prognostic factor.
The survival period of patients carrying MET F/SVs is significantly shortened, and they should be considered as a candidate group for upgraded management and targeted therapy.
With the popularization of genetic testing technology and the improvement of the accessibility of targeted drugs, the diagnosis and treatment of glioma are shifting from “one - size - fits - all” to “tailor - made.” Understanding the molecular characteristics of one's tumor may be the first step in winning this battle.
References
[1] Neuro Oncol. 2021;23:1231-1251.
[2] Cancer Discov. 2015;5:850-859.
[3] Cell. 2018;175:1665-1678.
[4] Genome Res. 2014;24:1765-1773.
Statement: This article is only for sharing. If it involves copyright and other issues, please contact us as soon as possible and we will correct it as soon as possible. Thank you!




 闽公网安备35021202000745
闽公网安备35021202000745