SpaceGen sequencing service facilitates research regarding the treatment of adult gastric isolated Langerhans cell histi
TThe Chinese Journal of Pathology published a short treatise titled "Clinical Pathology Analysis of 3 Cases of Adult Gastric Isolated Langerhans Cell Histiocytosis", which attracted widespread attention. It mainly discusses the clinical manifestations, pathological characteristics, immunophenotypes, molecular pathology, differential diagnosis, and treatment prognosis of adult gastric isolated Langerhans cell histiocytosis (LCH).
SpaceGen provided high - throughput sequencing technology for this study to detect and analyze hot mutation regions of BRAF, KRAS, and NRAS genes. It is once again confirmed that SpaceGen has leading and reliable quality control and testing capabilities, which can provide a strong guarantee for accurate clinical diagnosis and treatment.
The following is a quote from the article:
Clinical Pathology Analysis of 3 Cases of Isolated Langerhans Cell Histiocytosis in Adult Stomach
Chen Xi¹, Yuan Jingping¹, Zhao Lina¹, He Huihua¹, Ao Qilin², Ju Xianli¹, Chen Fangfang¹
¹Department of Pathology, People's Hospital of Wuhan University, Wuhan 430060, China;
²Department of Pathology, Tongji Hospital, Tongji School of Medicine, Huazhong University of Science and Technology, Wuhan 430060, China; Institute, Wuhan 430030, China
Corresponding author: Chen Fangfang, Email: 951333059@qq.com
Summary
Objective: To explore the clinical manifestations, pathological characteristics, immunophenotypes, molecular pathology, differential diagnosis, and treatment prognosis of adult gastric isolated Langerhans cell histiocytosis (LCH).
Methods: Analyze the clinicopathological characteristics, immunophenotypes, and molecular pathology of 3 cases of gastric isolated LCH, and summarize the relevant literature.
Results:
Case 1: A 23 - year - old male. Gastroscopy revealed a polyp - like bulge in the anterior wall of the junction of the gastric antrum and body.
Case 2: A 22 - year - old male. Gastroscopy found a bulge and erosive foci on the lesser curvature side of the gastric body.
Case 3: A 46 - year - old male. Gastroscopy detected a congestive erosion in the upper part of the gastric body, with a polyp in the middle.
Under the microscope, abnormal hyperplasia of Langerhans cells was observed in all 3 cases. The cells were of medium size, with slightly eosinophilic cytoplasm, oval nuclei, and visible nuclear grooves. There was a large number of inflammatory cells infiltrating in the background. Immunohistochemistry showed that CD1a, Langerin, and S - 100 proteins were all positive. Mutations in the BRAF V600E gene were detected in Cases 2 and 3. Thoracic and abdominal CT or positron emission computer tomography (PET - CT) were performed on all 3 cases to exclude LCH in other systems.
Conclusion: Gastric isolated LCH is very rare and has a good prognosis. Inexperienced doctors often do not fully understand the key points of diagnosing this disease. Attention should be paid to the differentiation from poorly differentiated adenocarcinoma to avoid misdiagnosis as a malignant tumor.
Fund projects: Wuhan City Science and Technology Plan Project (2017060201010172); Wuhan University People's Hospital Guidance Fund (RMYD2018M27)
Langerhans cell histiocytosis (LCH) is a rare disease characterized by the abnormal proliferation of Langerhans cells [1]. It mostly occurs in children and often involves multiple systems [2]. Gastric isolated LCH that occurs in adults is rare and has not been fully studied. In this study, 3 cases of adult gastric isolated LCH were collected, and the relevant literature was reviewed to discuss the clinicopathological characteristics, immunophenotypes, molecular pathology, and differential diagnosis of adult gastric isolated LCH. The purpose is to improve the understanding of the disease among clinicians and pathologists.
1. Information and methods
1) Case data: Collect the clinicopathological data of 3 patients diagnosed with adult gastric isolated LCH from the Department of Pathology of Wuhan University People's Hospital and the Institute of Pathology of Tongji Hospital Affiliated to Tongji School of Medicine of Huazhong University of Science and Technology from December 2017 to December 2022. Analyze their histological morphology, immunophenotypes, molecular pathology, and conduct follow - up.
2) Methods: The specimens were fixed with 3.7% neutral formaldehyde, followed by conventional dehydration, paraffin embedding, 4 - µm - thick slicing, HE staining, and observation under an optical microscope. Immunohistochemistry was performed using the EnVision method. The antibodies used were purchased from Dako company, including CD1a, Langerin, S - 100 protein, cyclin D1, Ki - 67, broad - spectrum cytokeratin (CKpan), CD56, chromogranin A (CgA), synaptophysin, p53, CD20, CD79a, CD3, CD30, HMB45, Melan A, and SOX10, all of which are ready - to - use antibodies. High - throughput sequencing technology was used to detect the hot mutation areas of the BRAF, KRAS, and NRAS genes. The detection platform was a DA8600 second - generation sequencer, and Xiamen SpaceGen Technology Co., Ltd. provided the sequencing services.
2. Result
1) Clinical data:
Example 1: A 23 - year - old male was admitted to the hospital for treatment due to “chest pain, 18 days after thoracic closed drainage”. Gastroscopy revealed that a polyp - like bulge could be seen in the anterior wall of the junction of the gastrointestinal sinus. The size was about 0.3 cm × 0.3 cm, with a smooth surface, a clear boundary, and a soft texture, but no obvious abnormalities were observed.
Example 2: A 22 - year - old male was admitted to the hospital for treatment due to “hunger pain for 3 days”. Gastroscopy showed that there was a bulge and erosive foci on the side of the small curvature of the stomach, which was soft in texture, but no obvious abnormalities were found.
Example 3: A 46 - year - old male was admitted to the hospital due to “intermittent upper abdominal distension for 2 months”. Gastroscopy showed a congestive and erosive focus near the cardia in the upper part of the stomach, and a polyp of 0.3 cm × 0.3 cm in size was seen on the large curvature side of the middle section. There was no obvious abnormality.
2) Pathological examination:
(1) General examination: In Example 1, a piece of gray - white tissue with a diameter of 0.2 cm was obtained. In Example 2, a piece of gray - white tissue with a diameter of 0.3 cm was obtained. In Example 3, 2 pieces of gray - white tissue with a diameter of 0.2 - 0.3 cm were obtained.
(2) Histological characteristics: All three cases showed abnormal hyperplasia of Langerhans cell nest - like distribution in the lamina propria of the gastric mucosa (Figure 1). The cells were medium - sized, with slightly eosinophilic cytoplasm, oval nuclei, unclear nucleoli, and nuclear grooves could be seen. Inflammatory cells such as eosinophils, lymphocytes, and plasma cells were observed in the background (Figure 2).
(3) Immunohistochemistry: In all three cases, CD1a (Figure 3), Langerin (Figure 4), and S - 100 protein were all positive (Figure 5). The Ki - 67 positive index was about 20% (Figure 6), and CKpan was negative. In Example 1, CgA, CD56, synapsin, CD20, CD79a, CD3, CD30, HMB45, Melan A, and SOX10 were all negative; in Example 2, CyclinD1 (Figure 7) was positive, synapsin, CD56, CgA were negative, and p53 individual cells were positive (Figure 8); in Example 3, cyclin D1 was positive, and p53 individual cells were positive.
(4) Genetic testing: Mutations in the BRAF V600E gene were detected in the primary foci of cases 2 and 3, while the KRAS and NRAS genes were not mutated.
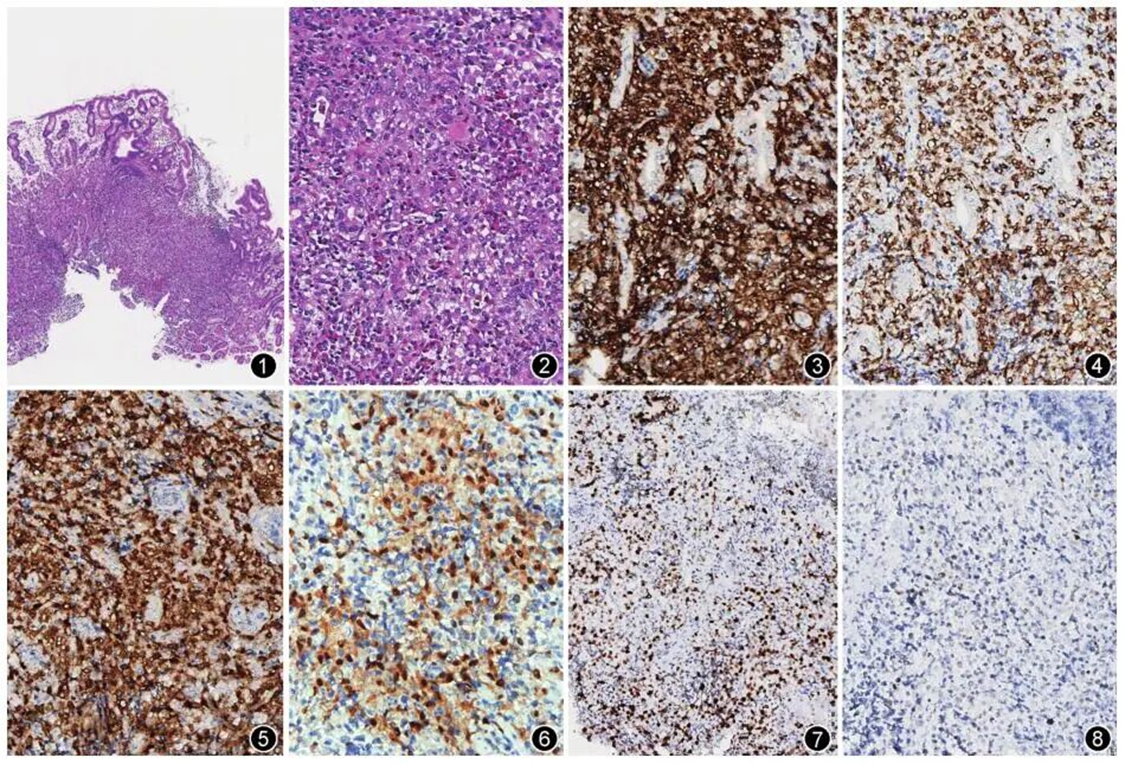
Figure 1: Gastric isolated Langerhans cell histiocytosis, low - magnification HE shows a denser layer of gastric mucosa around tumor cells.
Figure 2: Tumor cells show a typical coffee - bean - like nucleus, with a large number of inflammatory cells infiltrating the background. Medium - magnification HE.
Figure 3: Langerhans cell CD1a positive, EnVision method magnification.
Figure 4: Langerhans cell Langerin - positive, EnVision method magnification.
Figure 5: Langerhans cell S - 100 protein - positive, EnVision method magnification.
Figure 6: Langerhans cell Ki - 67 positive index about 20%, EnVision method magnification.
Figure 7: Langerhans cell cyclin D1 positive, EnVision method high - magnification.
Figure 8: Langerhans cell p53 individual cell positive, EnVision method medium - magnification.
3) Other examinations:
In Examples 1 and 3, postoperative review of chest and abdominal CT scans showed no obvious abnormalities. In Example 2, a bone marrow examination showed no tumor cells, and no genetic mutations were detected in the bone marrow. A whole - body positron emission computer tomography (PET - CT) showed no obvious abnormalities. All three cases excluded the involvement of other systems and were isolated.
4) Treatment and follow - up:
The patients in cases 1 and 3 were treated conservatively after biopsy. So far, the follow - up has been in good condition. In Example 2, the patient underwent gastroscopy review and endoscopic submucosal dissection (ESD) treatment after biopsy. During the operation, an erosive focus of 1.2 cm × 0.8 cm in size was found on the small curvature of the stomach. All the tissues tested after the operation were from tumor cells, and the follow - up period was from 6 months to 5 years. The patients are in good condition.
3. Discussion
In the past, LCH was considered to be three independent diseases: eosinophilic granuloma of bone, Letterer - Siwe disease, and Schüller - Christian disease. Since the three have many commonalities in clinicopathological characteristics, Lichtenstein[3] collectively referred to them as histiocytoproliferative disease X in 1953. In 1973, some scholars discovered that histiocytosis X is characterized by the abnormal local or systemic proliferation and spread of Langerhans cells. Therefore, in 1987, the collaborative group of the International Tissue Cell Association recommended that histiocytosis X be renamed LCH[4 - 5].
The scope of LCH lesions is variable, and it is clinically divided into three categories: single - foci single - system, multi - foci single - system, and multi - foci multi - system[6]. Among them, adult LCH that occurs solely in the stomach is extremely rare. We retrieved 15 cases from the Wanfang and PubMed databases[6 - 13]. The patients were aged 28 to 68 years old, and the ratio of men to women was 8:7. Among them, 8 cases were asymptomatic, and 7 patients had gastrointestinal symptoms, including stomach discomfort (3 cases), upper abdominal pain (2 cases), dysphagia (1 case), nausea/vomiting (1 case), etc. Isolated bulges or polyps (size 0.2 - 1.0 cm) were observed under gastroscopy in 10 cases, 1 case had multiple polyps, other lesions included erosion (2 cases) and ulcers (1 case), and only 1 case had no lesions. It is more common in the stomach (7 cases), and other parts include the gastrointestinal sinus (4 cases), the bottom of the stomach (1 case), and the angle of the stomach (1 case). There was also 1 case with lesions in the stomach and at the bottom of the stomach, and 1 case with lesions diffusely distributed throughout the stomach. In this group of cases, case 1 had no corresponding gastrointestinal symptoms, case 2 presented as hunger pain, case 3 presented as upper abdominal distension, and the three cases were all male, aged 22, 23, and 46 years old. Under gastroscopy, they all had isolated polyps or bulges located in the stomach. The clinical characteristics of the three cases are similar to those reported in the past, but the age of onset is younger, broadening the age range of their onset.
Some studies have proposed that the main points for the diagnosis of LCH are: it has specific microscopic manifestations and related clinical/imaging manifestations, and at least two immunohistochemical assays, CD1a and Langerin, are performed [14]. Histologically, LCH is characterized by the abnormally proliferating Langerhans cell nest - like distribution, medium - sized cells, round or oval nuclei, inconspicuous nucleolus, nuclear membrane folds, coffee - bean - like nuclear grooves, and slightly eosinophilic cytoplasm[15]. Reactive inflammatory cells such as eosinophils, lymphocytes, and plasma cells are often infiltrated in the background. In immunohistochemistry, tumor cells specifically express CD1a, S - 100 protein, and Langerin. Another study on skin LCH found that cyclin D1 was strongly expressed in the nucleus of tumor cells in 92.3% to 100% of cases, with a positive rate of 5% to 70%, while cyclin D1 was negatively expressed in reactive lesions[16 - 18]. This shows that cyclin D1 can be used to identify LCH and reactive lesions. At present, there are no research reports on the positivity rate of cyclin D1 in gastric isolated LCH, and there is no uniform standard for the positive interpretation of cyclin D1. In this group, the cyclin D1 positivity rates of Example 2 and Example 3 were 10% and 30%, respectively. In addition, in a recent study, it is recommended to perform systemic fluorodeoxyglucose PET - CT on LCH patients, including remote limbs, to help determine the scope and extent of the disease. The microscopic characteristics, immunohistochemistry, and imaging results of the three cases in this group all meet the above requirements.
The molecular characteristics that adult LCH may have include: (1) BRAF V600E mutation; (2) other activation mutations in the RAS - RAF - MEK - ERK pathway; (3) activation of kinase fusion[14]. Among them, BRAF V600E mutations exist in more than half of LCH cases[18]. Of the 15 cases retrieved, only 6 were tested for BRAF V600E mutations, of which 5 showed mutations. In this group, examples 2 and 3 used high - throughput sequencing technology to detect mutations in the BRAF V600E gene, but there were no mutations in the KRAS and NRAS genes. Combining the previous cases and the two cases in this group, the mutation ratio of BRAF V600E in adult gastric isolated LCH is 7/8, and more data need to be further studied.
The differential diagnosis of this disease is: (1) Erdheim - Chester disease: Microscopically, it shows mild yellow granulomatous inflammation and varying degrees of fibrosis, often strongly expressing factor aa and CD163, but not expressing S - 100 protein; (2) Rosai - Dofman disease: tumor cells lack nuclear grooves and express S - 100 protein and CD163; (3) Low - differentiation cancer: tumor cells are atypical, nuclear division is common, and CKpan is expressed; (4) Lymphoma: Tumor cells express corresponding markers of B cells and T cells, but do not express S - 100 protein. None of the above four diseases express CD1a and Langerin. It should be noted that the Ki - 67 positivity index of LCH has changed greatly. The positivity index of the three cases in this group is about 20%, while the previously reported cases can reach up to 60%[8]. Avoid misdiagnosis as a malignant tumor.
The prognosis of LCH is closely related to the number of affected organs, the number of dangerous organs involved, and the onset of the disease [19]. Patients with single - focal and single - system LCH generally only need local resection or observation. In the past, 15 cases of isolated LCH in the stomach were surgically removed (2 cases), treated with ESD (4 cases), or biopsied (9 cases). The follow - up time was from 3 months to 5.5 years, and the patients were in good condition during the follow - up period. In this group, cases 1 and 3 were observed after biopsy, case 2 patients underwent biopsy and ESD treatment, and the 3 patients were followed up for 6 months to 5 years. The condition was good, which is consistent with the treatment methods and prognosis reported in the past.
As can be seen from the above, adult gastric isolated LCH is rare. Most patients have no symptoms or have mild gastrointestinal symptoms, and most of them have isolated polyps or bulges under gastroscopy. Microscopic characteristics, immunohistochemistry, and systemic PET - CT to exclude other system involvement and molecular pathology suggesting mutations in the BRAF V600E gene are necessary for the diagnosis of this disease. Treatment is generally local resection or biopsy observation, and the overall prognosis is good. Due to the microscopic characteristics and large changes in the Ki - 67 positive index, misdiagnosis as low - differentiation cancer and other malignant tumors should be avoided.
References
[1] Andión Catalán M, Ruano Domínguez D, Azorí Cuadrillero D, et al. Gastrointestinal involvement in Langerhans cell histiocytosis[J].An Pediatr (Barc), 2015,83(4):279‑280. DOI: 10.1016/j. anpedi. 2015.04.014.
[2] Yoon HS, Lee JH, Michlitsch J, et al. Langerhans cell histiocytosis of the gastrointestinal tract: evidence for risk organ status[J]. J Pediatr, 2019, 212:66‑72. e3. DOI:10.1016/j. jpeds. 2019.05.003.
[3] Lichtenstein L. Histiocytosis X; integration of eosinophilic granuloma of bone, Letterer‑Siwe disease, and Schüller‑Christian disease as related manifestations of a single nosologic entity[J]. AMA Arch Pathol, 1953, 56(1):84‑102.
[4] Lamert F. Langerhans cell histiocytosis. Historical perspectives[J]. Hematol Oncol Clin North Am, 1998,12(2):213‑219. DOI: 10.1016/s0889‑8588(05)70506‑2.
[5] Berry DH, Becton DL. Natural history of histiocytosis‑X[J]. Hematol Oncol Clin North Am, 1987, 1(1):23‑34.
[6] Chen Hongmei, Ye Lin, Zhu Liangjun, etc. A case of gastric isolated Langerhans cell histiocytosis [J]. Journal of Clinical and Experimental Pathology, 2018, 34(9):1028‑1029. DOI: 10.13315/j. cnki. cjcep. 2018.09.021.
[7] Wang L, Yang F, Ding Y, et al. Gastrointestinal Langerhans cell histiocytosis with unifocal, single‑system involvement in adults: cases report and literature review[J]. J Clin Lab Anal, 2022, 36(12):e24765. DOI: 10.1002/jcla. 24765.
[8] Zhou Xiaoli, Fan Li, Gu Wenxian, etc. A case of gastric Langerhans cell histiocytosis [J]. Chinese Journal of Pathology, 2022, 51(10): 1062‑1064. DOI: 10.3760/cma.j.cn112151‑20220319‑00201.
[9] Wu Lihua, Li Chenfan, Zheng Yueping. A case of gastric isolated Langerhans cell histiocytosis [J]. Chinese Clinical Case Results Database, 2022, 04(01).
[10] Matsuoka Y, Iemura Y, Fujimoto M, et al. Upper 55 Chinese Journal of Pathology, September 2023, Volume 52, Issue 9 Chin J Pathol, September 2023, Vol. 52, No. 9Gastrointestinal Langerhans cell histiocytosis: a report of 2 adult cases and a literature review[J]. Int J Surg Pathol, 2021, 29(5):550‑556. DOI: 10.1177/1066896920964566.
[11] Li Ya'nan, Shao Shihong, Zhao Han, etc. A case of isolated Langerhans cell histiocytosis in the stomach [J]. Chinese Journal of Pathology, 2020, 49(6): 631‑633. DOI: 10.3760/cma.j.cn112151‑20191008‑00545.
[12] Yan F, Zhou Q, Gao Y, et al. Isolated Langerhans cell histiocytosis of the stomach: a case report and literature review[J]. Int J Clin Exp Pathol, 2018, 11(12):5962‑5968.
[13] Lee SJ, Hwang CS, Huh GY, et al. Gastric Langerhans cell histiocytosis: case report and review of the literature[J]. JPathol Transl Med, 2015, 49(5):421‑423. DOI: 10.4132/jptm. 2015.05.19.
[14] Goyal G, Tazi A, Go RS, et al. International expert consensus recommendations for the diagnosis and treatment of Langerhans cell histiocytosis in adults[J]. Blood, 2022, 139(17): 2601‑2621. DOI: 10.1182/blood. 2021014343.
[15] Emile JF, Abla O, Fraitag S, et al. Revised classification of histiocytoses and neoplasms of the macrophage‑dendritic cell lineages[J]. Blood, 2016, 127(22): 2672‑2681. DOI:10.1182/blood‑2016‑01‑690636.
[16] Shanmugam V, Craig JW, Hornick JL, et al. Cyclin D1 is expressed in neoplastic cells of Langerhans histiocytosis but not reactive Langerhans cell proliferations[J]. Am J Surg Pathol, 2017, 41(10):1390‑1396. DOI: 10.1097/PAS. 0000000000000897.
[17] Chatterjee D, Vishwajeet V, Saikia UN, et al. CyclinD1 is useful to differentiate Langerhans cell histiocytosis from reactive Langerhans cells[J]. Am J Dermatopathol, 2019,41(3):188‑192. DOI: 10.1097/DAD. 0000000000001250.
[18] Ben Rejeb S, Charfi L, Sahraoui G, et al. Cyclin D1:potential utility as marker for Langerhans cell histiocytosis[J]. J Immunoassay Immunochem, 2021,42(4):370‑379. DOI: 10.1080/15321819.2020.1870132.
[19] Selway JL, Harikumar PE, Chu A, et al. Genetic homogeneity of adult Langerhans cell histiocytosis lesions: Insights from BRAFV600E mutations in adult populations[J]. Oncol Lett, 2017, 14(4):4449‑4454. DOI:10.3892/ol. 2017.6774.
[20] Matsubara Y, Kobayashi M, Hijikata Y, et al. Gastrointestinal lesion in adult‑onset Langerhans cell histiocytosis[J]. Int J Clin Oncol, 2020, 25(11):1945‑1950.




 闽公网安备35021202000745
闽公网安备35021202000745