



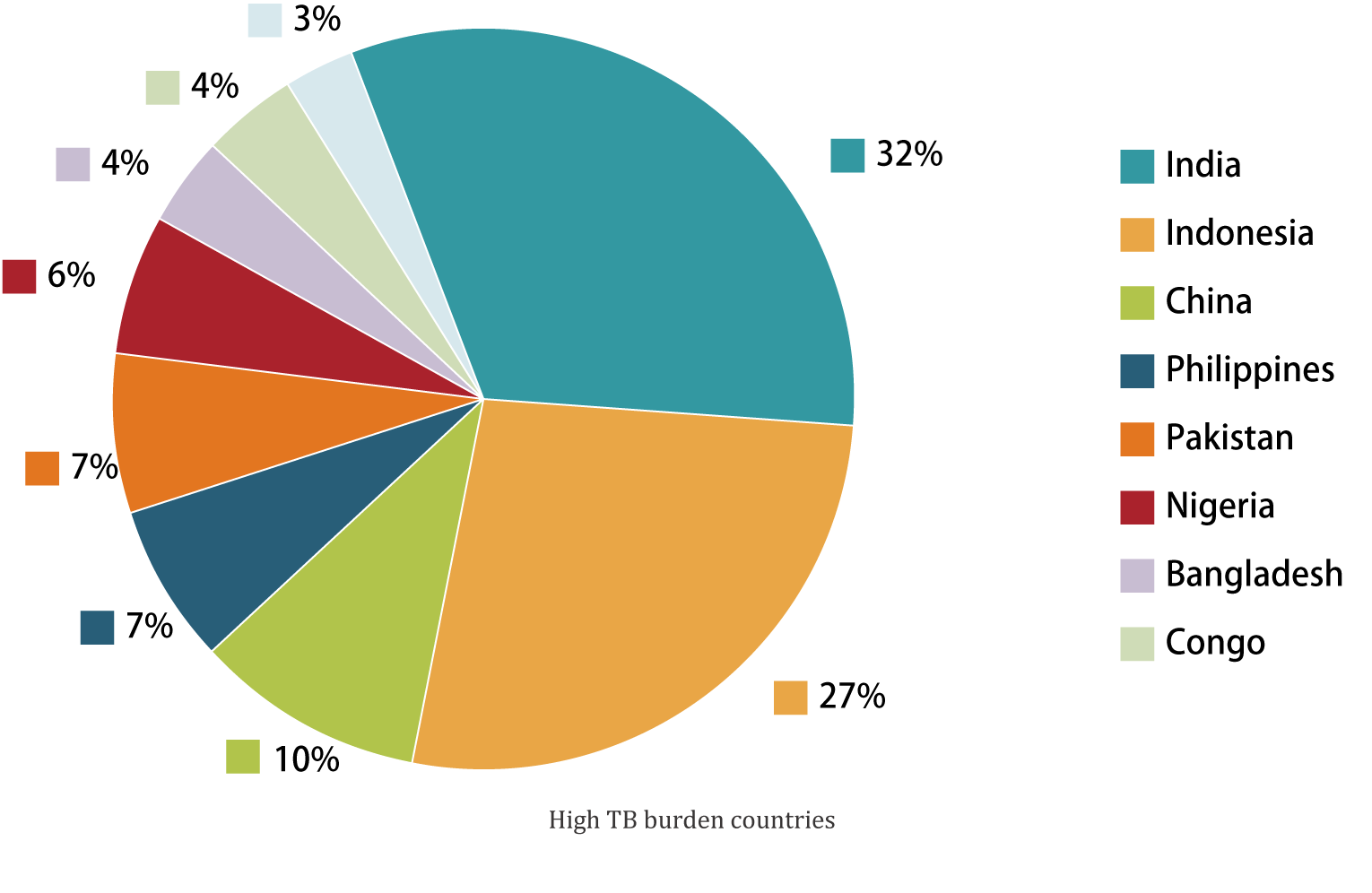
According to the World Health Organization's (WHO) Global Tuberculosis Report 2023, there are 10.6 million new cases of TB worldwide in 2022, and a total of 1.3 million TB-related deaths. China is one of the top three countries with a high burden of TB in the world, with an estimated 748,000 new cases of TB in 2022, accounting for 7.1% of the global new cases.
Tuberculosis (TB) and non-tuberculosis (NTB) caused by mycobacterium infection in High TB burden countries are serious infectious diseases that harm human health. They are prevalent in the world and are one of the key infectious diseases under global control. Because TB and non-TB symptoms are similar but treatment regimens are very different, most non-TB mycobacteria are naturally resistant to anti-TB drugs. Therefore, accurate detection of mycobacterium infection identification in lung tissue is essential for the diagnosis and treatment of the disease.

Matrix assisted laser desorption/ionization time of flight mass spectrometry (MALDI-TOF-MS),the combined molecules are ionized with the help of the matrix, and the ions are accelerated through the flight pipe under the action of an electric field. Their mass and charge can affect their flight time, so the detection objects are accurately separated and identified according to the different flight times when they arrive at the detector.
Accurate identification of common clinical mycobacterium tuberculosis complex groups and 26 pathogenic mycobacteria, clear pathogenic bacteria, to achieve individualized precision treatment.
1.Nucleic acid extraction
2.Template loading
3.Mass spectrometry detection
4.Data analysis
5.Report generation



 闽公网安备35021202000745
闽公网安备35021202000745