



Tuberculosis is one of the major infectious diseases that China focuses on controlling. The Chinese government has always attached great importance to the prevention and control of tuberculosis. In recent years, the Chinese government has issued documents such as the Outline of the Healthy China 2030 Plan, the Action Plan to Curb Tuberculosis (2019-2022) and the Further Strengthening of the Prevention and Control of drug-Resistant Tuberculosis. It is clearly proposed to further strengthen the prevention and control of tuberculosis and drug-resistant tuberculosis, and reduce the incidence and mortality of tuberculosis.
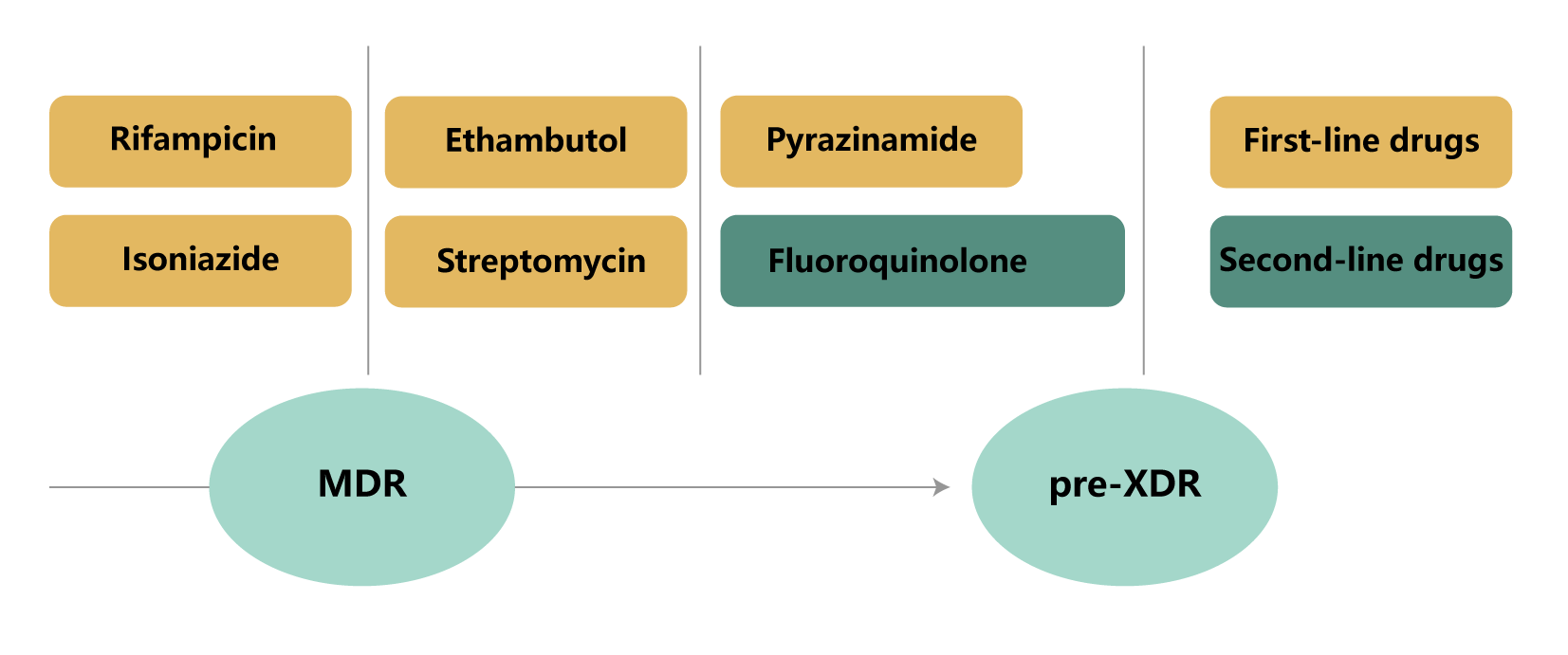
MDR-TB remains a global public health crisis, with an estimated 410,000 people living with MDR-TB or Rifampicin-resistant TB (MDR/RR-TB), and only about two in five people receiving treatment. The treatment success rate of MDR/RR-TB patients in China is only 51%, which is still far behind the global treatment success rate of 63%. At the same time, the high cost of diagnosis and treatment also makes it difficult for some patients with drug-resistant tuberculosis to bear, resulting in the delay of individual illness and even the spread of drug-resistant tuberculosis, making the tuberculosis prevention and control situation worse. Therefore, accurate detection of anti-TB drug resistance mutations is essential for the diagnosis and treatment of the disease.
Matrix assisted laser desorption/ionization time of flight mass spectrometry (MALDI-TOF-MS), the combined molecules are ionized with the help of the matrix, and the ions are accelerated through the flight pipe under the action of an electric field. Their mass and charge can affect their flight time, so the detection objects are accurately separated and identified according to the different flight times when they arrive at the detector.
Accurately identify and evaluate the drug resistance of 6 common first - and second-line anti-tuberculosis drugs, identify pathogenic bacteria and drug sensitivity, and achieve individualized precision treatment.
1.Nucleic acid extraction
2.Template loading
3.Mass spectrometry detection
4.Data analysis
5.Report generation



 闽公网安备35021202000745
闽公网安备35021202000745