



Epidermal growth factor receptor (EGFR), a receptor tyrosine kinase, is expressed in about 50% of patients with non-small cell lung cancer (NSCLC) [1] and is closely related to the occurrence and development of tumors. EGFR is the most frequently mutated driver gene in East Asian NSCLC patients, with a mutation frequency of 38.4%, among which the mutation frequency in East Asian female NSCLC patients is 51.1%[2]. But for Europeans or Americans, 19% of NSCLC patients carry EGFR mutations[3]. EGFR gene mutation is the most important predictor of the efficacy of EGFR kinase inhibitors (TKI) and is a prerequisite for clinical decision of whether a patient can be treated with EGFR-TKI. Both the National Comprehensive Cancer Network (NCCN) include EGFR mutation testing as a Class 1 recommendation ⁽⁴⁾.
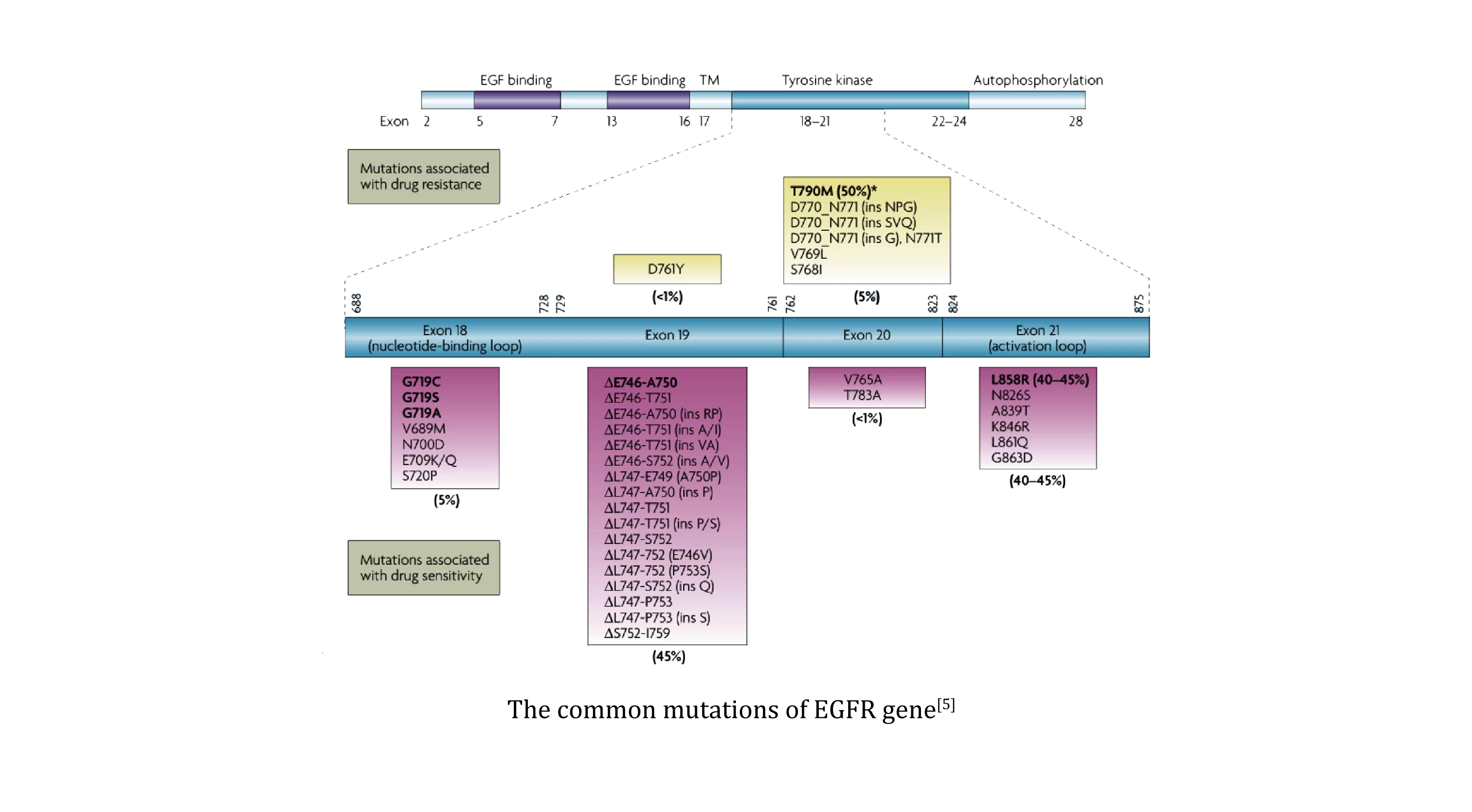
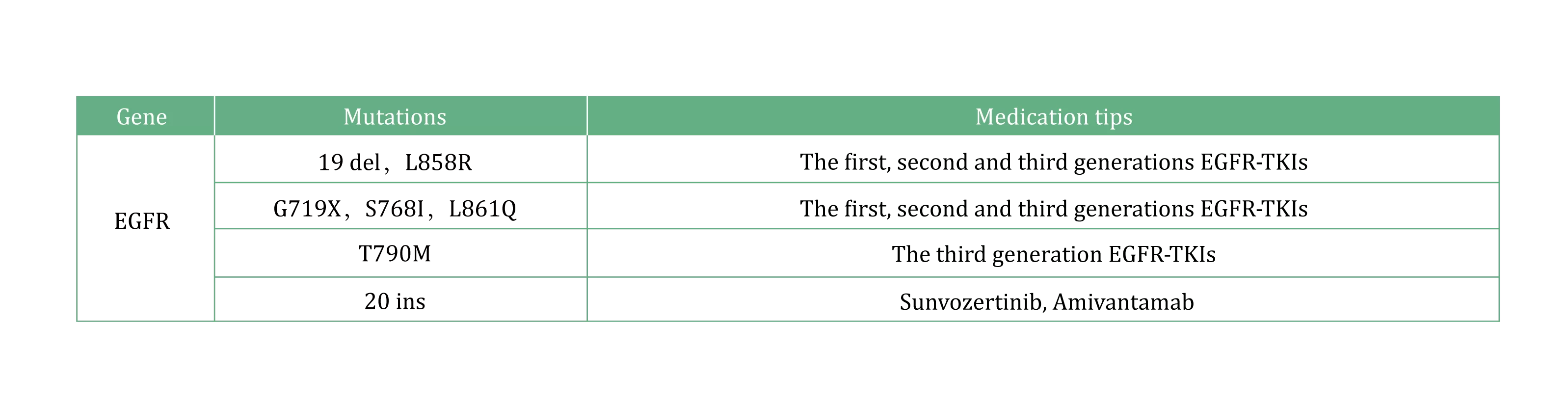
EGFR carcinogenic mutations occur mostly on exon 18-21, among which 19 del and L858R account for 85%-90% of all EGFR mutations [5], and are also the most common EGFR-TKI sensitive mutations. The T790M mutation suggests that the first and second generations of EGFR-TKI are resistant, and the third generation of EGFR-TKI needs to be used. 20 ins mutations are insensitive to conventional EGFR-TKI, suggesting the use of EGFR 20 ins inhibitors.
[1] Nat Rev Cancer. 2007 Mar;7(3):169-81.
[2] Oncotarget. 2016 Nov 29;7(48):78985-78993.
[3] World J Clin Oncol. 2021 Apr 24:12(4):217-237.
[4] NCCN NSCLC 2026 V3
[5] Nat Rev Cancer. 2007 Mar;7(3):169-81.
1.EGFR mutation detection in patients with operable stage IB-IIIA NSCLC to guide adjuvant targeted therapy.
2.EGFR gene mutation testing is performed for patients with inoperable stage III and IV NSCLC before first-line treatment, and treatment is guided by molecular classification.
3.In patients with EGFR-TKI resistance, biopsy is recommended for EGFR gene mutation detection.
1. Nucleic acid extraction
2. Set up qPCR
3. Amplification
4.Data analysis




 闽公网安备35021202000745
闽公网安备35021202000745