



Chronic myeloid leukemia (CML) accounts for 15%-20% of all leukemia, and more than 95% of CML patients have the characteristic BCR-ABL fusion gene. Tyrosine kinase inhibitors (TKIs) targeting BCR-ABL1, including Imatinib, Nilotinib, and Dasatinib, are effective in the treatment of CML, but their efficacy is limited in patients with primary or secondary resistance.
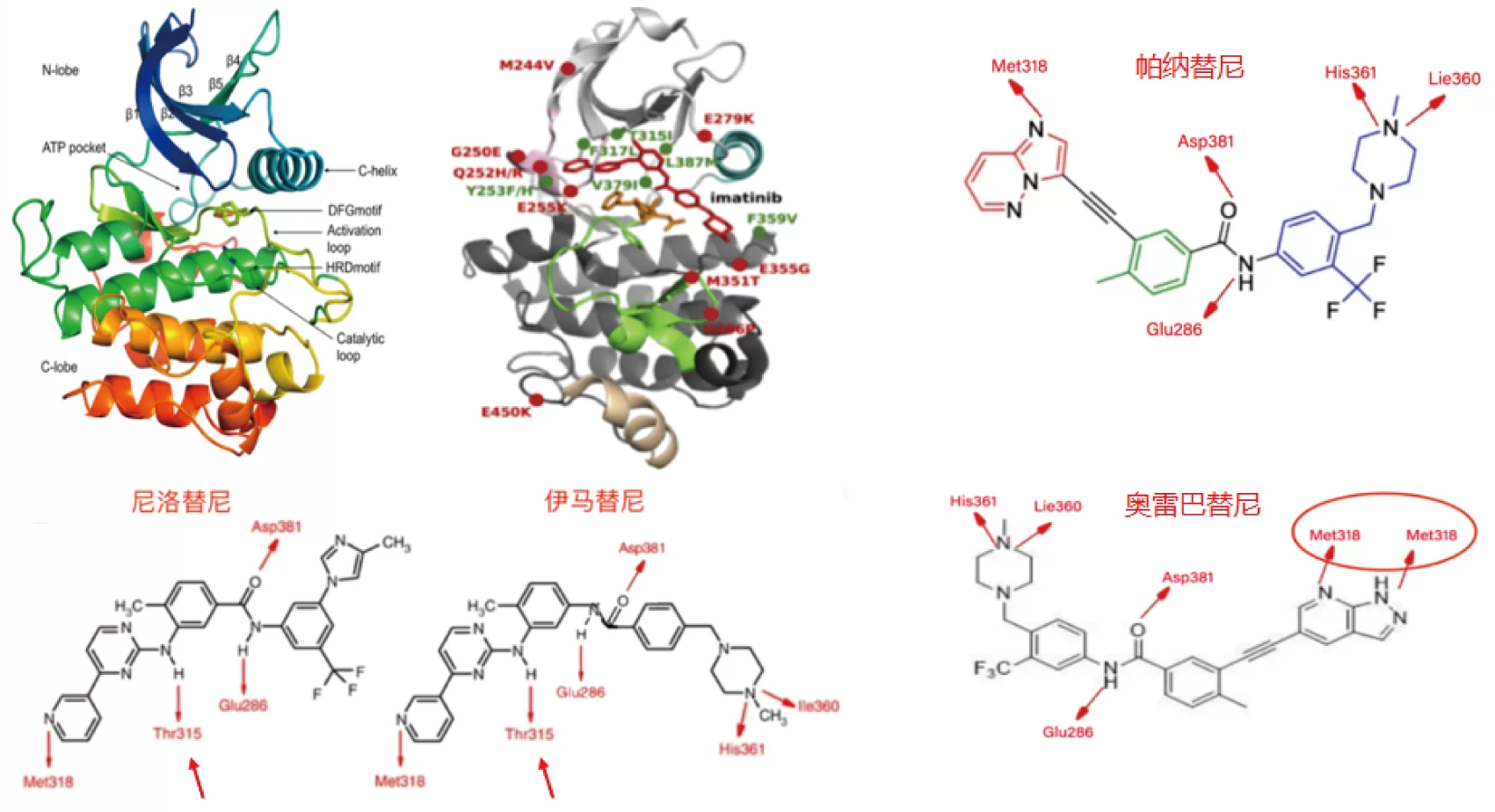
In the T315I mutation, threonine (Thr) at exon 6 of the ABL1 gene is replaced by isoleucine (Ile) at position 315, changing the base from ACT to ATT. After mutation, Ile315 cannot form hydrogen bonds with IM, and additional C-H bonds on the side chain of Ile after replacement will cause spatial interference, which is not good for drug binding. Therefore, T315I mutation is resistant to Imatinib, Nilotinib and Dasatinib. Therefore, the detection result of BCR-ABL gene T315I mutation is an important indicator to guide the medication of CML patients.
The NCCN CML Clinical Guideline states that Bosutinib, Dasatinib, and Nilotinib are prohibited for patients with T315I mutations, that Asciminib and Ponatinib are alternative therapies, and that the FDA approved Olverembatinib to enter Phase Ⅲ trials for global registration.

In leukemia patients with Imatinib, Nilotinib and Dasatinib resistant CML patients, BCR-ABL gene T315 mutation detection is performed before changing TKI drugs to provide reference for CML patients.
1.Nucleic Acid Extraction
2.Set up qPCR
3.Amplification
4.Data Analysis



 闽公网安备35021202000745
闽公网安备35021202000745