



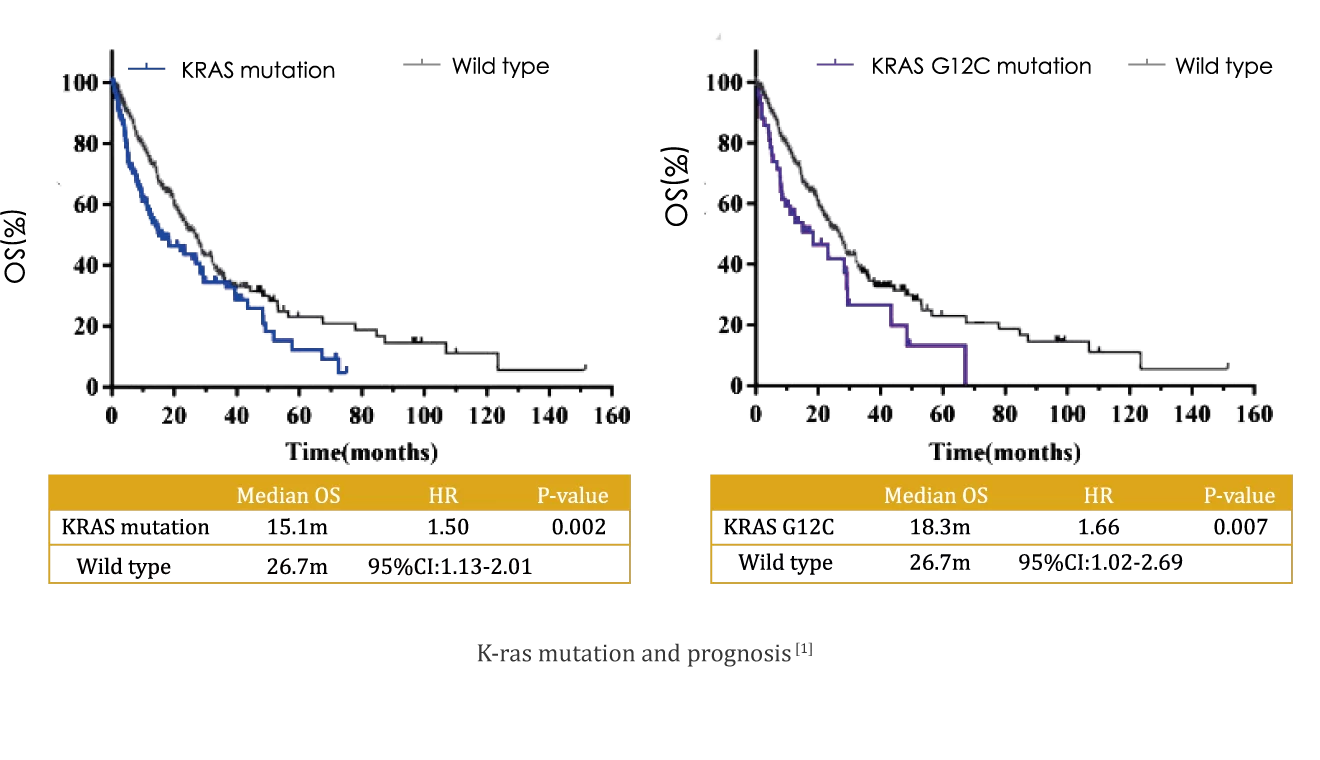
Kirsten rat sarcoma viral oncogene (K-ras) is the most frequently mutated gene in patients with non-small cell lung cancer (NSCLC) in USA/Europe and the second most frequently mutated gene in patients with NSCLC in East Asian, with a mutation frequency of 9.8%; about 30% of which are G12C mutations [1]. K-ras mutation is associated with poor prognosis (compared to wild type, HR=1.5, P=0.002) [1]. In May 2021, the FDA approved AMGEN's KRAS G12C inhibitor Sotorasib for the treatment of adult patients with KRAS G12C-mutated locally advanced or metastatic NSCLC, who have received at least one prior systemic therapy.
Cetuximab and panitumumab are monoclonal antibody drugs that target epidermal growth factor receptor (EGFR), which can competitively prevent EGFR from binding to its ligand and inhibit its downstream signal transmission. K-ras is located in the MAPK signaling pathway downstream of EGFR. The mutant K-ras protein does not depend on the regulation of EGFR, therefore, patients are resistant to cetuximab and panitumumab. The FDA specifically stated in the product instructions for cetuximab and Panitumumab that these drugs are not recommended for colorectal cancer patients with K-ras mutation.
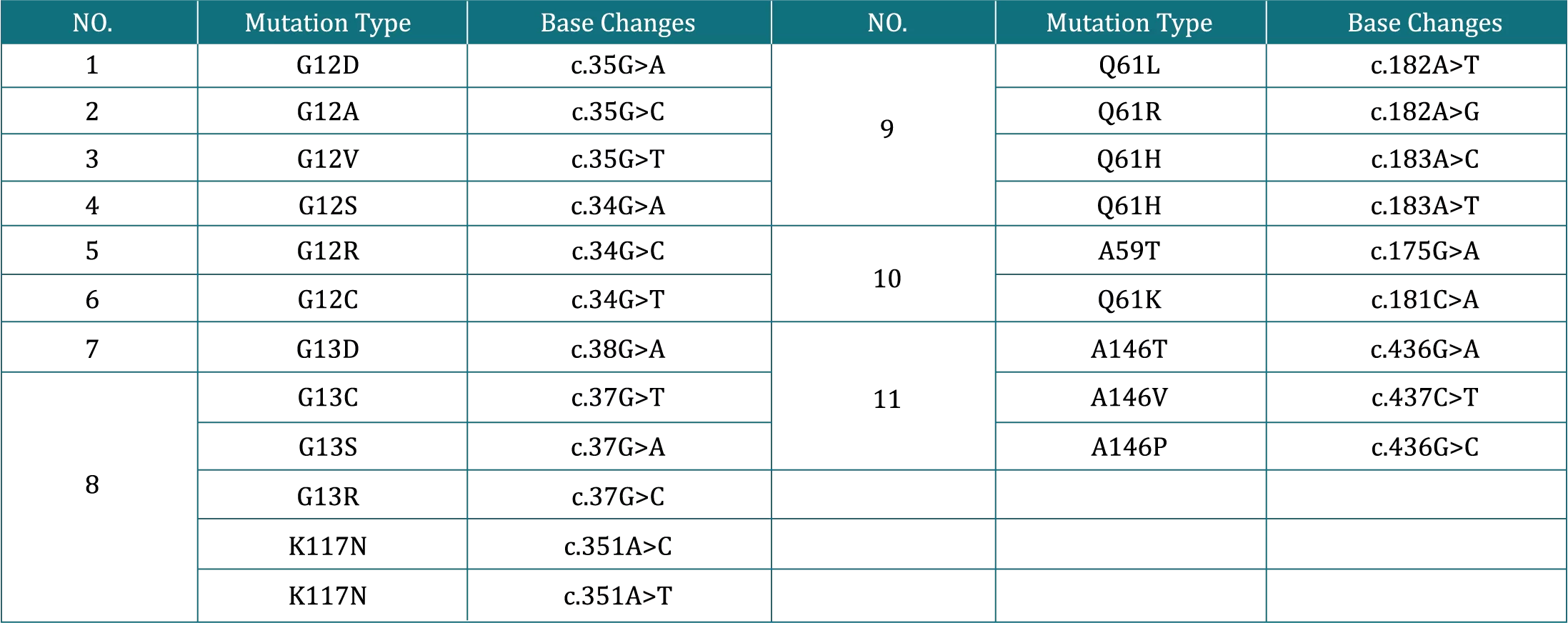
Select patients for treatment of locally advanced or metastatic NSCLC with G12C inhibitor based on the presence of KRAS G12C
mutation in tumor or plasma specimens.
Patients with mutant KRAS gene show poor response to anti-EGFR or anti-HER2 therapies.
1. Nucleic acid extraction
2. Set up qPCR
3. Amplification
4.Data analysis



 闽公网安备35021202000745
闽公网安备35021202000745