



Over the past 30 years, the incidence of thyroid carcinoma has continued to rise rapidly in many countries and regions around the world. In 2022, there will be nearly 800,000 new cases worldwide. Although the 5-year survival rate of thyroid cancer has gradually increased (to 84.3%), many patients still experience tumor recurrence or metastasis.
Gene diagnosis based on molecular changes of thyroid cancer has been used as an auxiliary tool for pathological diagnosis in clinical practice. The incidence of thyroid nodules shows an increasing trend year by year, of which 8 % to 16 % of nodules are malignant, and the main purpose of thyroid nodule evaluation is to identify benign and malignant. Studies have shown that RET gene rearrangement occurs most frequently in papillary thyroid carcinoma ( PTC ). Compared with the most common BRAF mutation in PTC, although the frequency of RET rearrangement in PTC is not very high, accounting for about 10 % -20 %, there is a mutual exclusion between RET rearrangement and BRAF mutation.
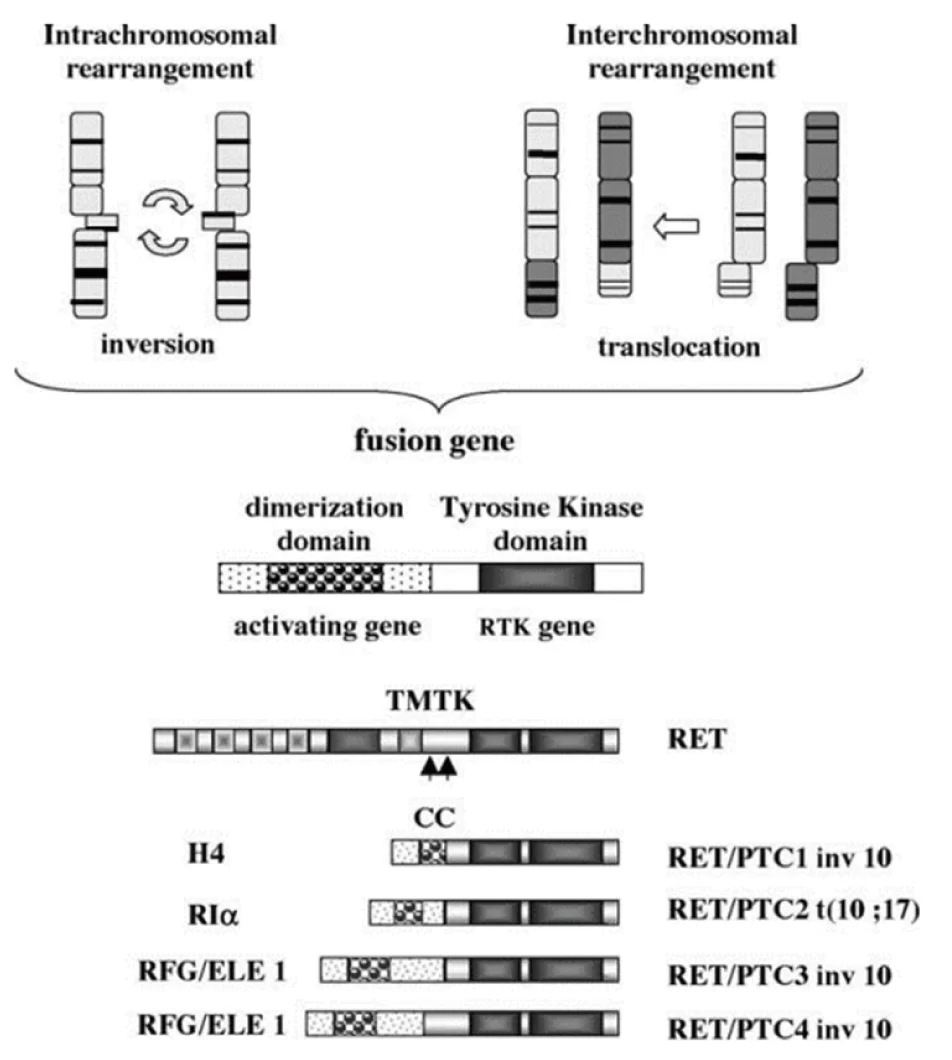
RET / PTC rearrangement is a highly specific marker for PTC diagnosis. Guidelines and expert consensus recommend : PTC with RET rearrangement is more malignant, and active surgical treatment is recommended as soon as possible. At the same time, RET gene is a therapeutic target for RET-driven malignant tumors, and targeted therapy is an important treatment for advanced PTC with RET gene rearrangement.
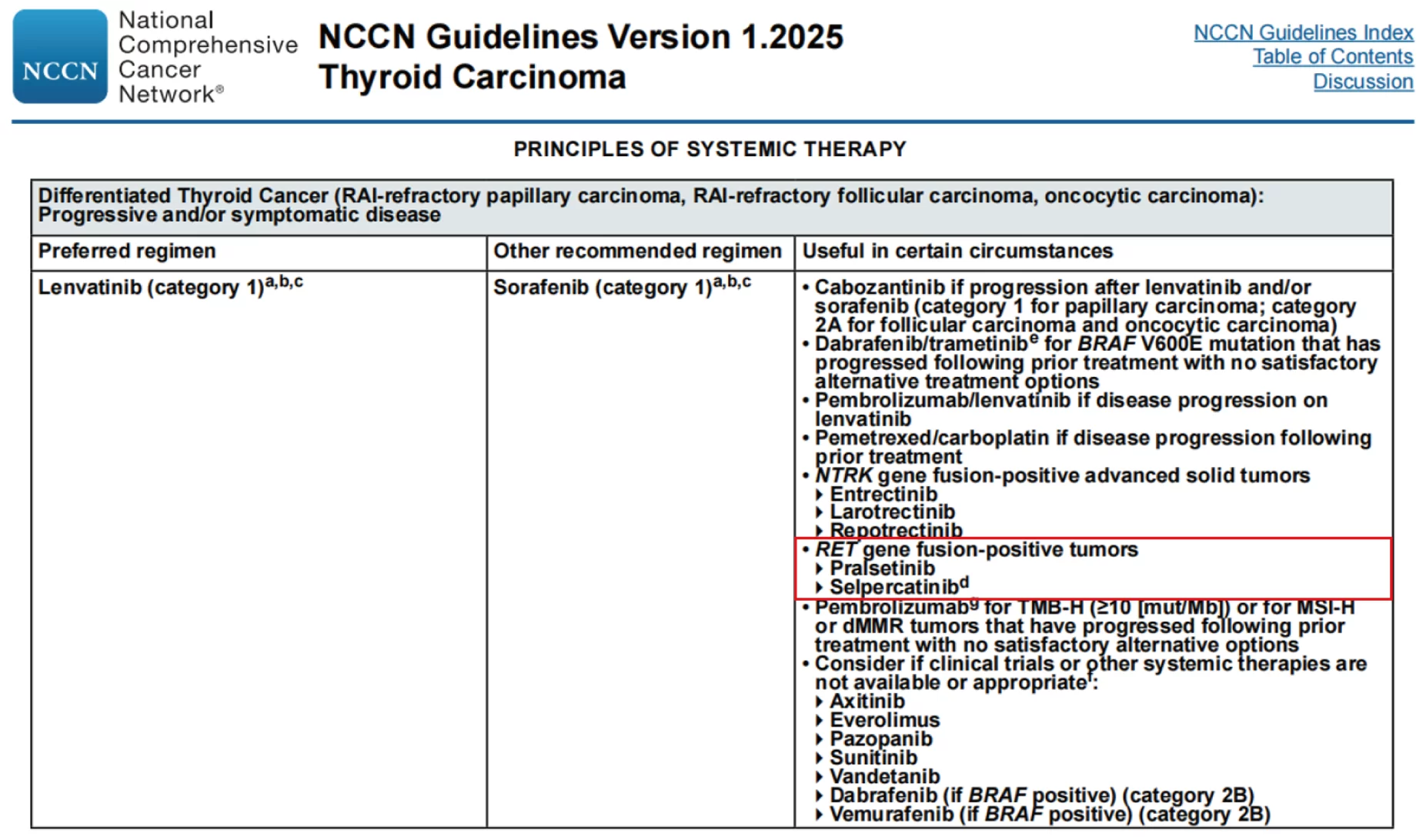

( 1 ) To assist the diagnosis of benign and malignant thyroid nodules and the classification of thyroid carcinoma.
( 2 ) Guiding targeted therapy
1. Patients with thyroid nodules with uncertain results diagnosed by US-FNA cytology ;
2. Patients with advanced thyroid carcinoma to be selected for targeted therapy ;
3. Patients with postoperative recurrence.
1. Nucleic acid extraction
2. Set up qPCR
3. Amplification
4.Data analysis



 闽公网安备35021202000745
闽公网安备35021202000745