



Cervical cancer is a malignant tumor that seriously threatens women's health. Early diagnosis and treatment are crucial in reducing cervical cancer mortality rates. Cervical intraepithelial neoplasia (CIN, original CIN1) can be classified into low-grade squamous intraepithelial lesion (LSIL, original CIN2 and CIN3) and high-grade lesions (HSIL). Timely detection and treatment of HSIL, which has the potential to progress to invasive cervical cancer, are the main objectives in the management of cervical cancer, aiming to minimize the risk of invasive cervical cancer development.
Cervical/vaginal cytological smear examination and HPV testing are currently the primary screening methods for detecting early-stage cervical cancer and precancerous lesions (CIN). Cytological examination relies on the subjective judgment of pathologists, making it prone to underdiagnosis or overdiagnosis. Among abnormal cell smears, atypical squamous cells of undetermined significance (ASC-US) account for over 50%, yet the probability of biopsy-confirmed CIN2/3 diagnosis is below 10%. While HPV testing demonstrates high sensitivity, 80% of HPV infections in women are transient, with fewer than 10% progressing to CIN.
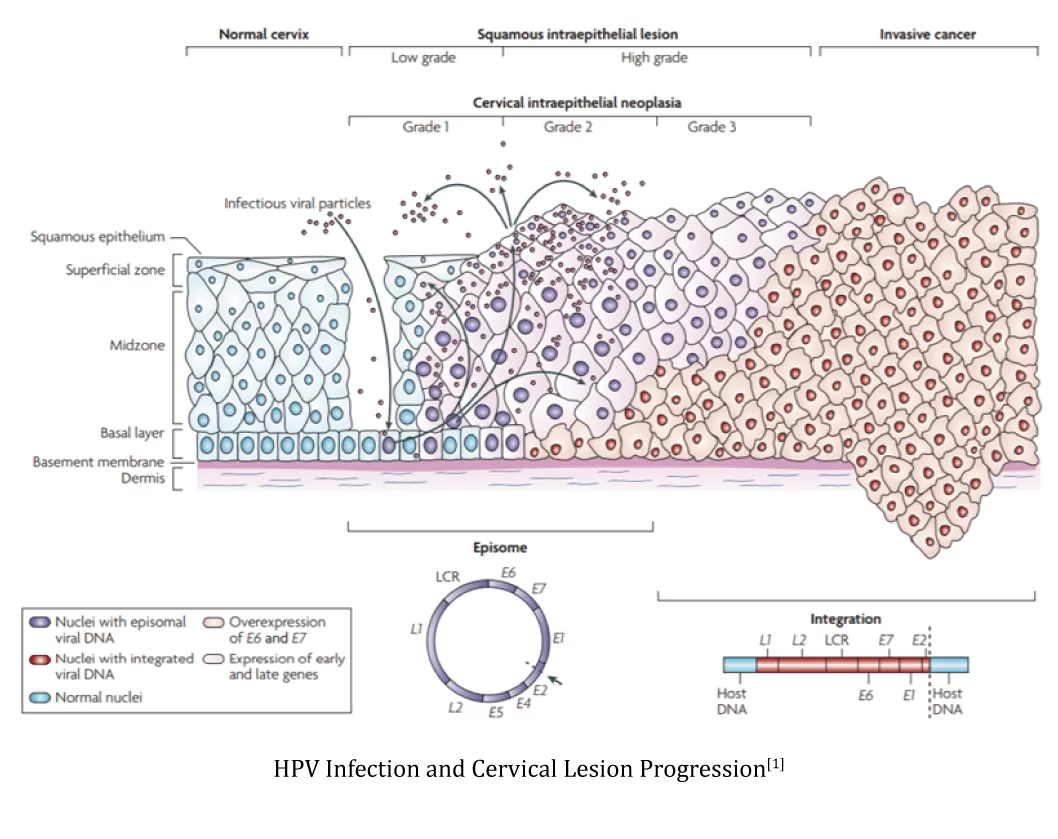
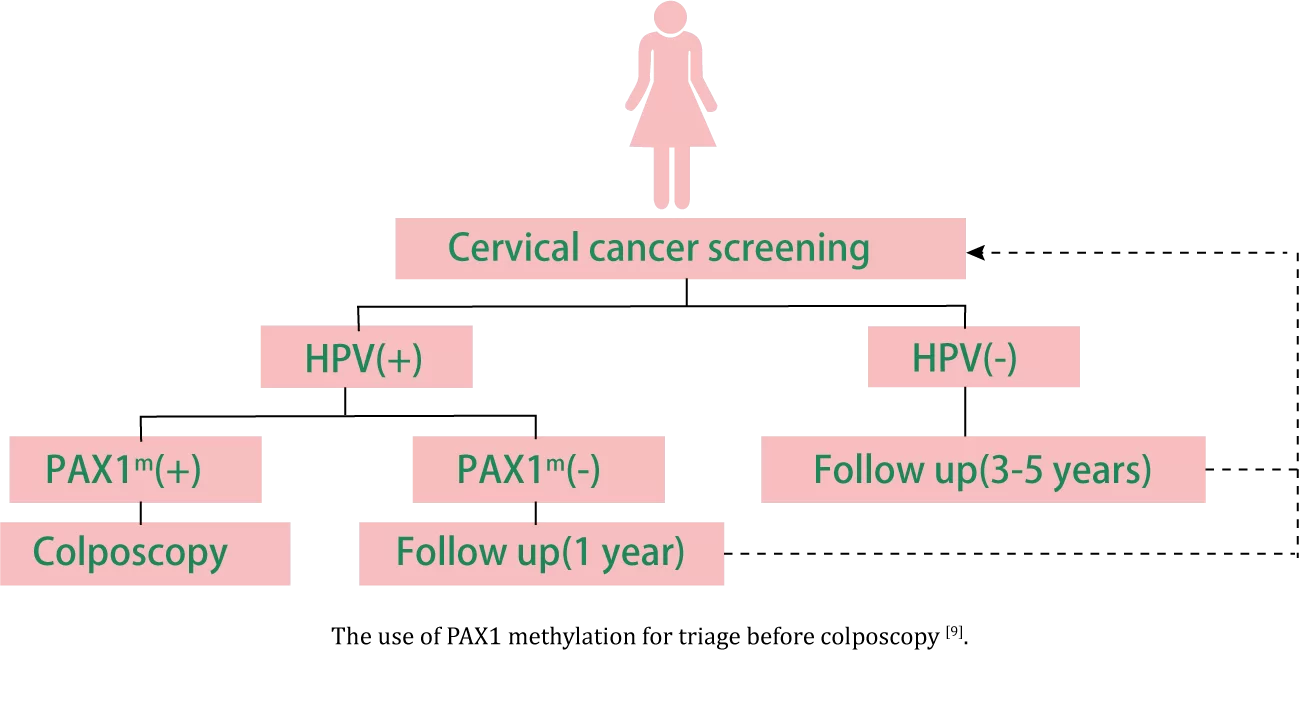
PAX1, a tumor suppressor gene, shows significant correlation between high methylation in its promoter region and cervical cancer[2]. The primary advantage of PAX1 methylation detection lies in its high specificity, with methylation levels demonstrating strong positive correlation with cytological grading and cervical lesion severity[3]. It can serve as a triage tool between positive HPV testing or ASC-US cytology results and colposcopy. PAX1 methylation testing can reduce unnecessary colposcopies and biopsies by 30%-60%[4,5], showing particularly notable efficacy in detecting type III transformation zones and cervical adenocarcinoma[6,7]. Beyond triage application, PAX1 methylation detection combined with HPV testing can enhance cervical cancer primary screening sensitivity without compromising specificity[8].
[1] Nat Rev Cancer. 2007 Jan;7(1):11-22.
[2] Sci Rep. 2019 Jun 24; 9 (1): 9195.
[3] Cancer Manag Res. 2020; 12: 2567–2576.
[4] Int J Gynecol Cancer. 2014 Jun;24(5):928-34.
[5] Oncotarget. 2017 Jul 22; 8 (37): 62274-62285.
[6] Future Oncol. 2022 Jul;18(23):2583-2592.
[7] Int J Gynecol Cancer. 2014 Feb; 24 (2): 201-9.
[8] Int J Cancer. 2008 Jul 1;123(1):161-7.
[9] Mol Genet Med. 2019 Mar;7(3):e506.
1.Applicable to women aged 30 and above with positive non-16/18 hrHPV test results, aiding in identifying the need for colposcopy to achieve triage purposes;
2.Suitable for suspected patients with cytological abnormalities or symptomatic cases;
3.Can be combined with HPV testing for primary screening, enabling simultaneous detection with a single sample collection.
1. hrHPV-positive individuals, particularly those with non-16/18 hrHPV subtypes;
2. Patients with cytological results of ASC-US or higher-grade abnormalities;
3. Individuals with recurrent cervical inflammation, persistent infections, or unexplained vaginal bleeding;
4. General populations undergoing cervical cancer screening.
1、Nucleic Acid Extraction
2、Modification
3、Set up qPCR
4、Amplification
5、Data Analysis



 闽公网安备35021202000745
闽公网安备35021202000745