



Thyroid nodules can be found in various age groups. In the adult population, the incidence rate detected through physical examinations is approximately 5%-7%, while the incidence rate detected through ultrasound examination ranges from 20%-76%. Different pathological types of thyroid nodules (tumors) exhibit significant differences in their biological behavior. From benign thyroid adenomas and indeterminate thyroid nodules to thyroid carcinoma, these variations can have a profound impact on patient prognosis and treatment options.
AUXILIARY EXAMINATION AND DIAGNOSIS
Ultrasound: Ultrasound examination is simple and non-invasive, with high specificity and sensitivity for thyroid nodule detection.
Cytopathology Diagnosis Report: The Bethesda System is used for the reporting of cytopathology diagnosis.
Molecular Testing: To improve the accuracy and effectiveness of thyroid fine-needle aspiration (FNA) cytology diagnosis, molecular testing is recommended for III and IV category thyroid FNA specimens or Puncture eluent.

NCCN Guidelines for Diagnosis and Treatment of Thyroid Carcinoma 2025.V1
Clinical Application of Gene Detection in Thyroid Carcinoma
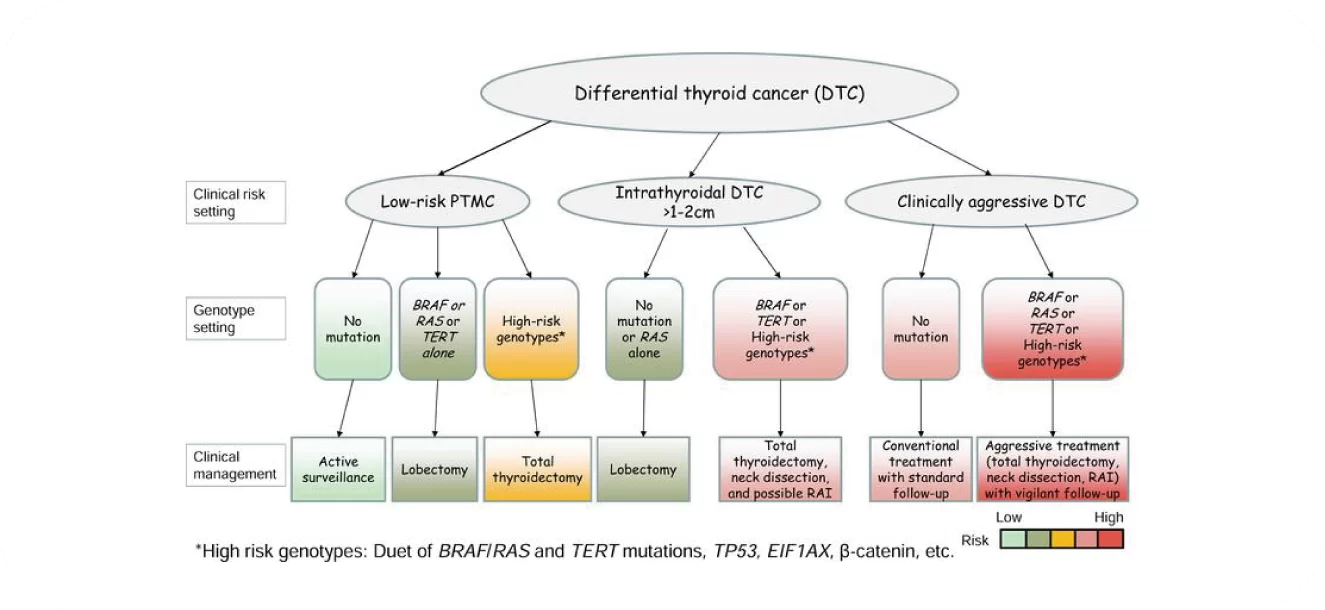
Endocrinol Metab Clin North Am, 2019, 48(1):109-124.
This test adopts the high-throughput sequencing method and covers the genes related to the identification, prognosis and adjuvant treatment of thyroid tumors recommended in the NCCN guidelines, CSCO guidelines and expert consensus.

1.Patients with thyroid nodules with uncertain results diagnosed by US-FNA cytology ;
2.Patients undergoing surgical treatment, radioactive iodine therapy or ablation ;
3.Patients with advanced thyroid carcinoma to be selected for targeted therapy ;
4.Patients with hereditary MTC background and their families.
( 1 ) To assist the diagnosis of benign and malignant thyroid nodules and the classification of thyroid carcinoma.
( 2 ) To guide the choice of surgical plan.
( 3 ) Guidance of radioactive iodine therapy
( 4 ) Guiding targeted therapy
( 5 ) Assessing genetic risk
1. Nucleic Acid Extraction
2. Library Preparation (3.5 hours total time)
3. Sequencing
4. Auto-data Analysis
5. Report





 闽公网安备35021202000745
闽公网安备35021202000745