



The susceptible genes for breast cancer, including BRCA1 and BRCA2, are important tumor suppressor genes that play a role in homologous recombination repair (HRR) of DNA damage. Mutations in the BRCA1/2 genes can lead to homologous recombination deficiency (HRD), resulting in a significant increase in genomic instability. The variant status of the BRCA1/2 genes is of significant importance in genetic risk assessment, treatment selection, and prognosis determination for related tumors such as ovarian cancer, breast cancer, pancreatic cancer, prostate cancer, etc. The BRCA1/2 genes have relatively long sequences and diverse forms of mutations, with mutation sites scattered throughout the entire length of both genes. Therefore, BRCA1/2 gene testing must simultaneously cover the coding regions and adjacent boundary regions (ideally within ±20 bp) [1]. Germline mutations in the BRCA1/2 genes originate in reproductive cells, significantly increasing the risk of developing breast cancer, ovarian cancer, and other related tumors [1]. About 10% of breast cancer patients [2-3], 10-15% of ovarian cancer patients [4], and 10% of prostate cancer patients [5] are caused by germline mutations in the BRCA1/2 genes.
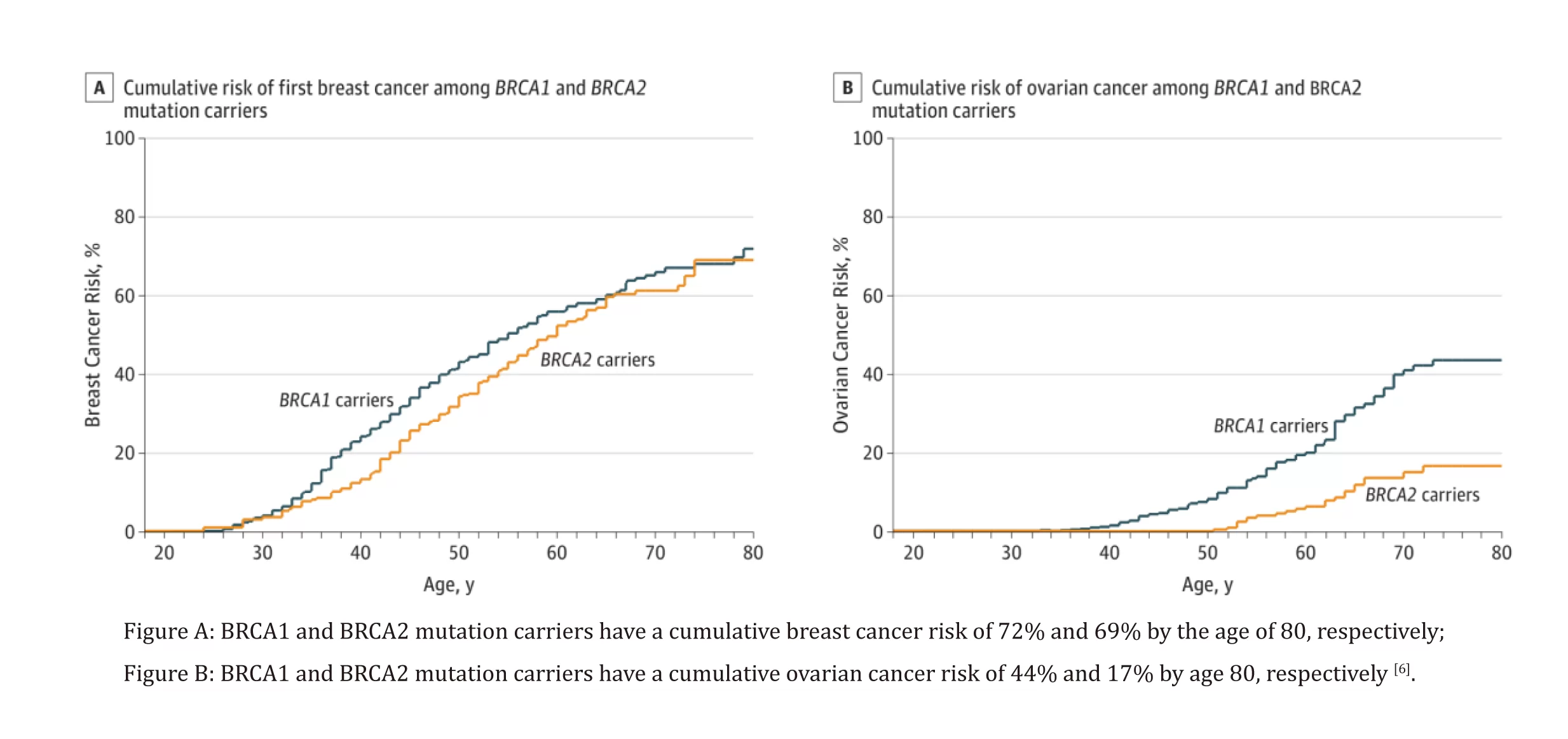
The variant status of BRCA1/2 genes is closely related to the effectiveness of Poly(ADP-ribose) polymerase (PARP) inhibitors. In recent years, the FDA and NMPA have approved a variety of PARP inhibitors for the treatment of related tumors [1]. Approximately 20% of ovarian cancer patients carry BRCA1/2 gene mutations [7], making them the population that benefits the most from maintenance treatment with PARP inhibitors. Furthermore, multiple PARP inhibitors have been approved for various indications in breast cancer, prostate cancer, and pancreatic cancer. These indications all require BRCA1/2 gene testing as a companion diagnostic.
References:
[1] BRCA1/2 Data Interpretation Chinese Expert Consensus (2021 Edition)
[2] Chinese Expert Consensus on Clinical Diagnosis and Treatment of Hereditary Tumors (2021 Edition) - Hereditary Breast Cancer
[3] Int J Cancer. 2017 Jul 1; 141(1): 129-142.
[4] Chinese Expert Consensus on Clinical Diagnosis and Treatment of Hereditary Tumors (2021 Edition) - Hereditary Ovarian Cancer
[5] JCO Precis Oncol. 2017 Jul;2017:PO.17.00029.
[6] JAMA. 2017 Jun 20; 317(23): 2402-2416.
[7] Nature. 2011 Jun 29; 474(7353): 609-15.
Predict therapeutic efficacy of PARP inhibitors in patients with cancers such as ovarian cancer, breast cancer, prostate cancer , pancreatic cancer , etc.
Facilitate the diagnosis of hereditary cancer syndromes, including hereditary breast-ovarian cancer syndrome (HBOC) and hereditary prostate cancer.





 闽公网安备35021202000745
闽公网安备35021202000745