



Pan cancer driver gene mutation detection panel aims at the companion diagnostic genes approved by FDA and recommended by NCCN guidelines, covering 56 genes related to cancer treatment and prognosis, including 3000 cosmic mutation sites, so as to realize low-cost, high sensitivity and high-throughput gene detection of tumor tissue and circulating free DNA of cancer patients.Cancer is a complex polygenic disease caused by the gradual accumulation of gene mutations. When the genes regulating cell growth are mutated or damaged, the cells lose control and proliferate and differentiate disorderly and infinitely, leading to the occurrence of malignant tumors.Tumor precise diagnosis and treatment products provide important reference basis for precise drug treatment, molecular typing and efficacy evaluation by accurately analyzing the unique gene mutation information of each tumor patient.
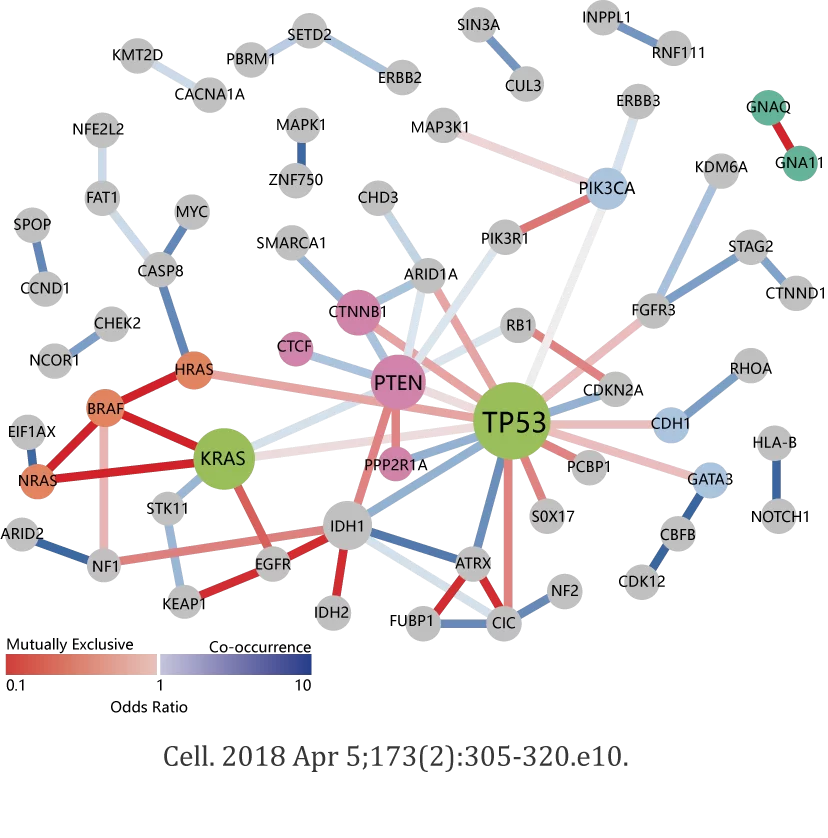
NCI-MATCH, also known as MATCH, is a precision medicine cancer treatment clinical trial. In this trial, people with cancer are assigned to receive treatment based on the genetic changes found in their tumors through genomic sequencing and other tests. Genomic sequencing is a laboratory method that is used to determine the genetic makeup of cancer cells. People whose tumors have genetic changes that match one of the treatments in the trial may receive that treatment if they meet other eligibility criteria. The trial seeks to determine whether treating cancer based on these specific genetic changes is effective, no matter the cancer type.
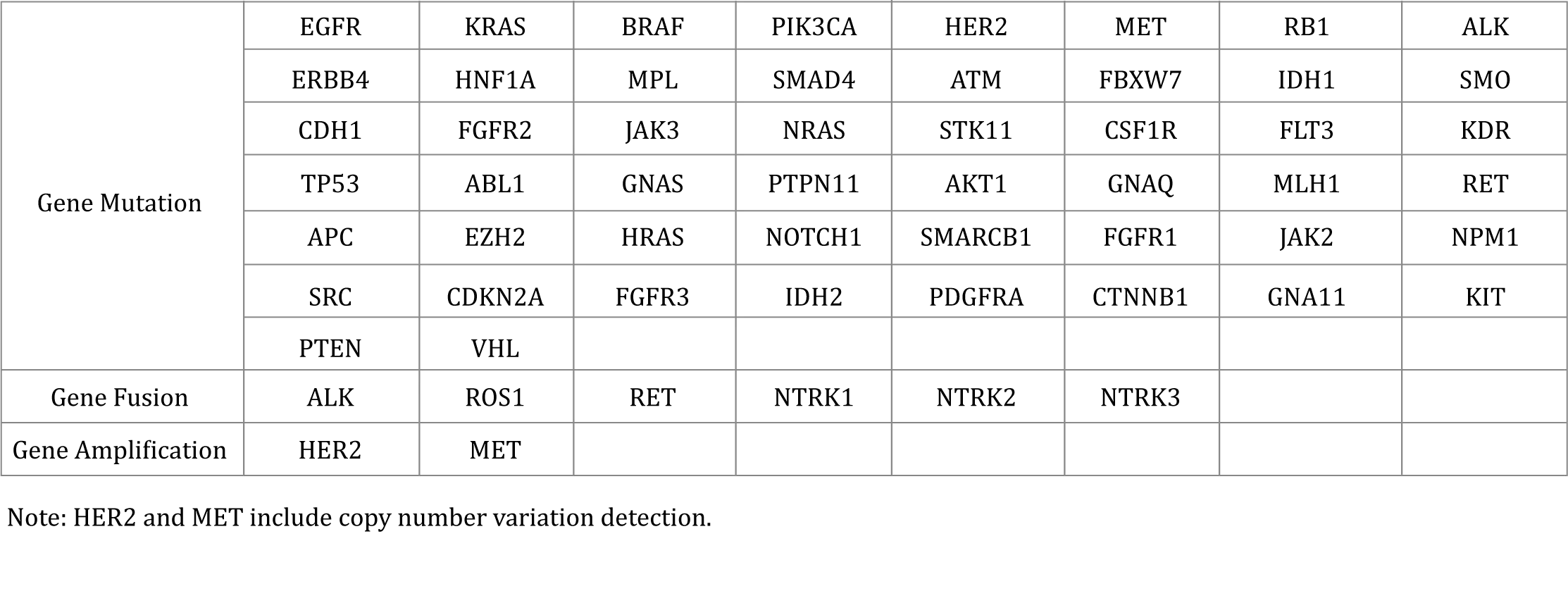
It is suitable for a variety of solid tumors and some hematological tumors. It is mainly used to evaluate the drug sensitivity and prognosis of all tumor patients who need targeted therapy, to assist clinicians in selecting appropriate targeted drugs, and to guide the choice of systematic treatment of patients more comprehensively. At the same time, some genetic related information can be provided to assess family genetic risk.
1. Nucleic Acid Extraction
2. Library Preparation (3.5 hours total time)
3. Sequencing
4. Auto-data Analysis
5. Report





 闽公网安备35021202000745
闽公网安备35021202000745