



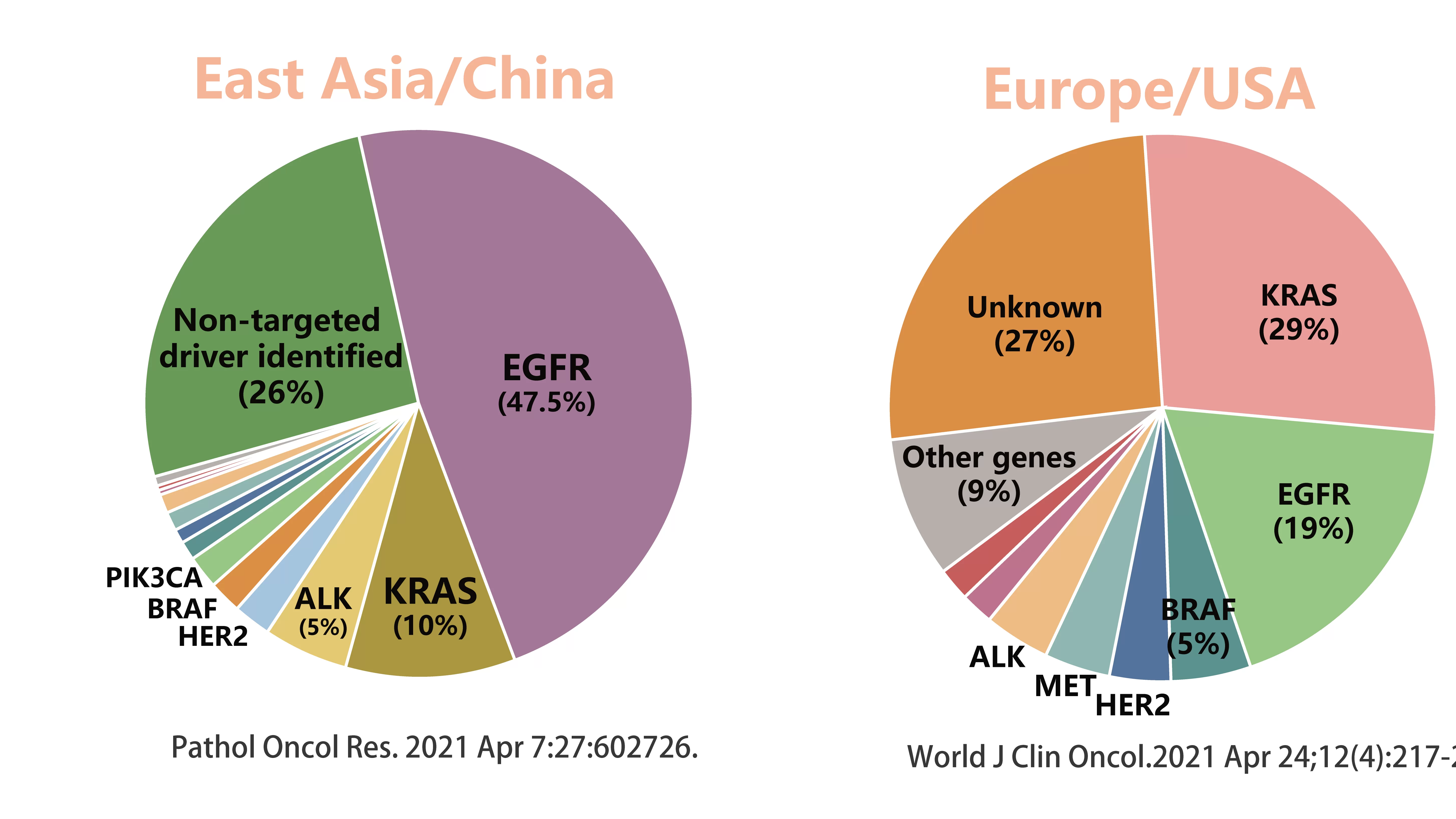
Tumor is the neoplasm of normal tissue, of which cancer is malignant neoplasm driven by various of tumorigenesis factors. Gene mutations caused by environment and heredity (including point mutation, deletion, insertion, copy number gain, gene fusion and so on) may lead to the division and growth of normal cells out of control and finally induce the formation of tumors. Targeted drugs are able to target the pathologic molecules in cancer cells caused by specific gene mutations, which will maximize the drug efficacy and minimize the side effects. Therefore, the detection of the patient's gene status can contribute the clinicians to select suitable target drugs. The occurrence of tumor often involves several or even a dozen oncogenes or cancer suppressor genes. The study showed that 47.5% of Asian patients with non-small cell lung cancer carried EGFR mutations, followed by KRAS at 10%. For Europeans or Americans, 29% of NSCLC patients carry KRAS mutations, and only 19% have EGFR mutations. In addition, common non-small cell lung cancer driver genes include ALK, BRAF, ROS1, RET, MET, HER2, etc.
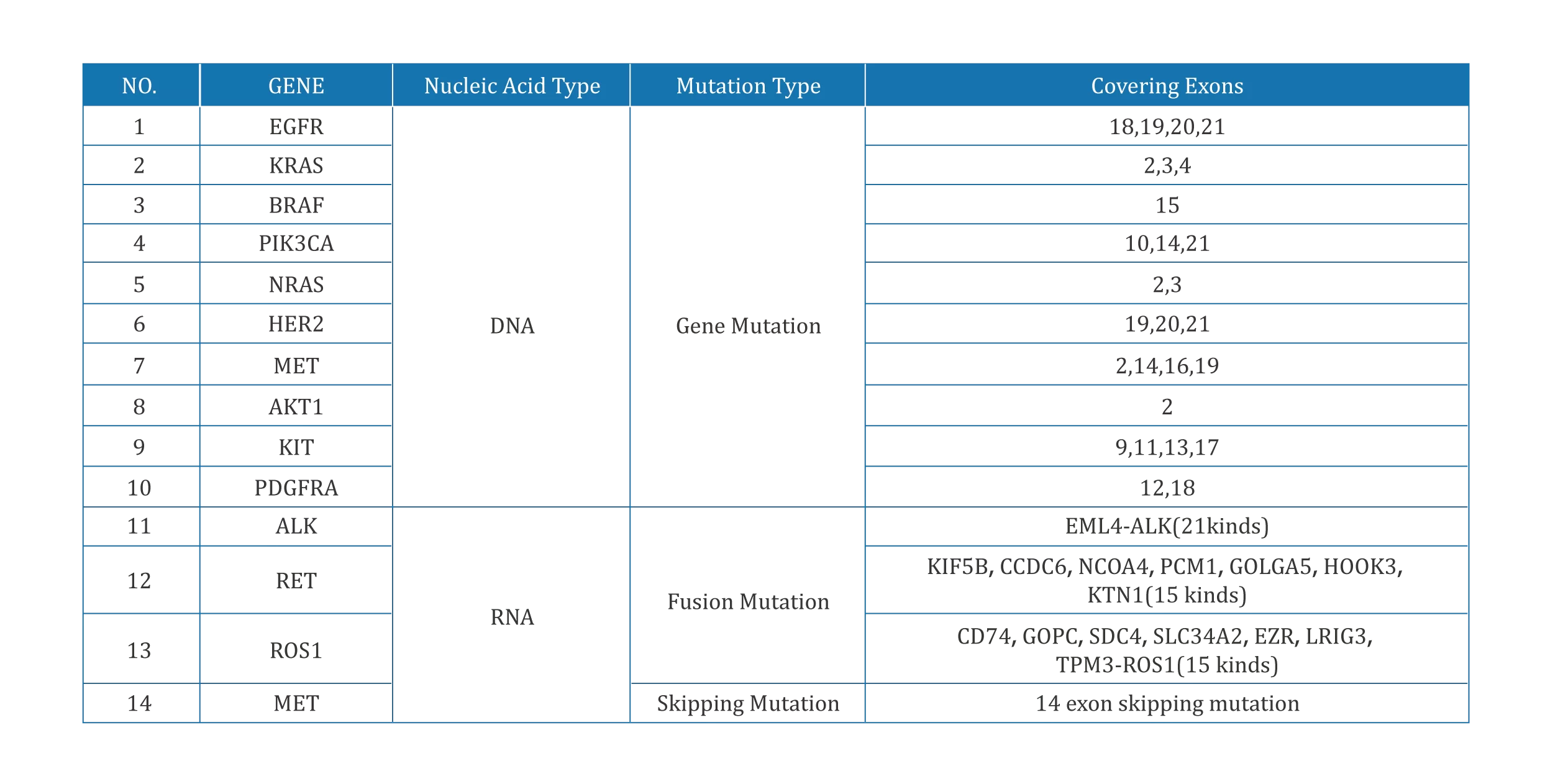
Personalized treatment:Patients with non-small cell lung cancer can undergo genetic testing before using targeted drugs to assist clinicians to judge the sensitivity of patients to drugs;
Monitoring drug efficacy and resistance: Patients who are resistant to targeted drug therapy and need to adjust their medication regimen.
This clinical trial adopts a blinded comparative trial design, and the FDA-approved listing of Thermo Fisher Scientific Oncomine DX Target Test(NGS)

1. Nucleic Acid Extraction
2. Library Preparation (3.5 hours total time)
3. Sequencing
4. Auto-data Analysis
5. Report





 闽公网安备35021202000745
闽公网安备35021202000745