




Gliomas originate from glial cells of the brain and are the most common primary intracranial tumors. Glioblastoma is the most aggressive tumor of the central nervous system (CNS). It accounts for the majority (58.4%) of gliomas and overall survival is as low as 12-15 months. According to the global incidence and death of cancer in 2022, the annual new cases of brain and central nervous system cancer in the world is 321,000, and the annual death cases are 248,000. According to the fifth edition of the World Health Organization (WHO) classification of central nervous system tumors in 2021, brain gliomas are divided into five groups according to histological and molecular pathological characteristics, namely: adult diffuse glioma, childhood diffuse LGG, childhood diffuse HGG, localized astrocytoma, and ependymal tumors. With the development of pathology and the advancement of pathology detection technology, especially the improvement of omics technology such as second-generation sequencing and DNA methylation profile, the genetic background and occurrence and development mechanism of brain gliomas are gradually becoming clear. More and more molecular markers have been shown to play an important role in the classification, classification, grading, prognosis and treatment of brain gliomas.
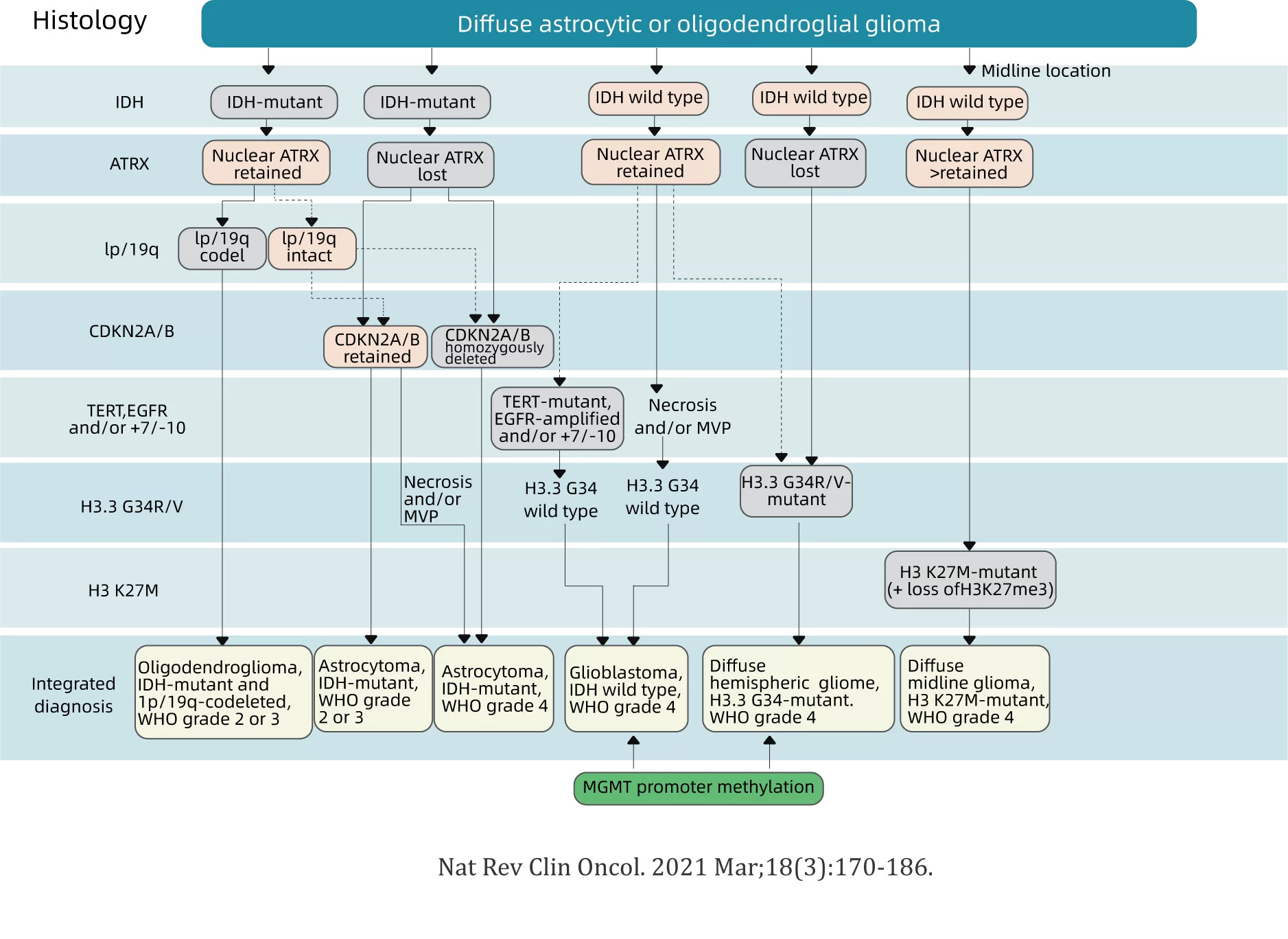
Assist to classification: The diagnosis of brain glioma requires the acquisition of specimens by tumor resection or biopsy, tissue and molecular pathology examination, and the determination of pathological grade and molecular classification. At present, the main molecular pathological markers include: IDH1/2 mutation, chromosome 1p/19q joint deletion, +7/-10, TERT mutation, etc.
Prognosis evaluation: Different molecular classifications and markers may have different prognostic characteristics. IDH mutants are associated with a better prognosis, and gliomas lacking homozygous CDKN2A and/or CDKN2B are associated with a worse prognosis.
Treatment guidance: Molecular testing for glioblastoma is encouraged, if driver mutations (such as the BRAFV600E mutation or NTRK fusion) are detected, targeted therapy can be performed, and patients may have more treatment options in clinical trials.
1. Nucleic Acid Extraction
2. Library Preparation (3.5 hours total time)
3. Sequencing
4. Auto-data Analysis
5. Report




 闽公网安备35021202000745
闽公网安备35021202000745