



Endometrial carcinoma is the most common malignant tumor of the female reproductive system in developed countries. The prognosis of endometrial carcinoma varies greatly, and the classical Bokhman's dualistic model and histological classification have certain limitations. In 2013, the Cancer Genome Atlas ( TCGA ) used a multi-omics approach to classify endometrial carcinoma into four subtypes[1]. Molecular classification has important guiding significance for the prognosis and individualized treatment of patients with different subtypes.

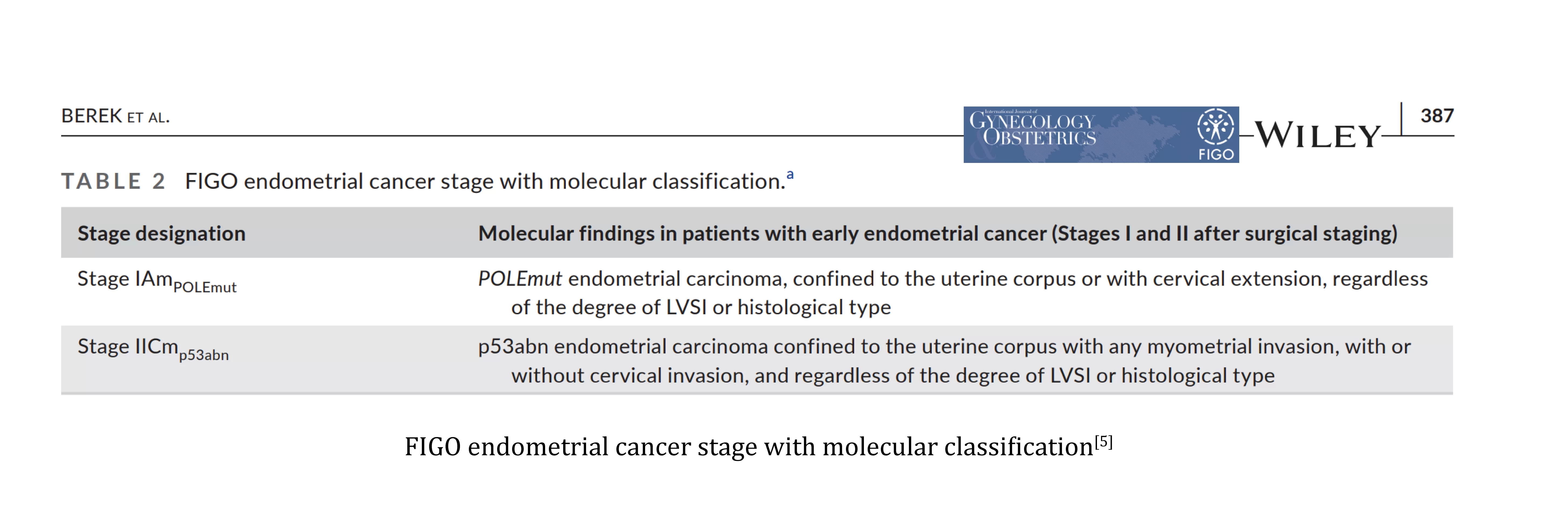
NCCN, WHO, CSCO and other guidelines recommend molecular classification of all diagnosed endometrial cancer patients[2-4].The 2023 edition of FIGO endometrial cancer staging encourages complete molecular classification in all endometrial cancers for prognostic stratification, adjustment of staging based on molecular classification, and as a potential influencing factor for adjuvant or systemic treatment decisions[5].
With the continuous development of research and application, there are also some reports on the internal heterogeneity of different molecular subtypes : pathogenic mutations in exon 3 of CTNNB1 gene are associated with poor prognosis of non-specific molecular subtype ( NSMP ) [6] ; MSI-H / dMMR patients with MLH1 methylation have poor prognosis and immunotherapy response[7-8]. The prognosis of serous carcinoma, clear cell carcinoma and carcinosarcoma is different[9-10]. The existing molecular classification methods are not perfect. The addition of more auxiliary typing genes can more effectively indicate the prognosis of patients with endometrial cancer and guide treatment decisions.
[1] Nature. 2013 May 2; 497(7447):67-73.
[2] NCCN Uterine Neoplasms Guidelines 2026 v2
[3] Geburtshilfe Frauenheilkd. 2021 Oct;81(10):1145-1153.
[4] CSCO Endometrial Cancer Diagnosis and Treatment Guidelines 2024
[5] Int J Gynaecol Obstet. 2023 Aug;162(2):383-394.
[6] Int J Gynecol Cancer. 2020 Dec;30(12): 2002-2007.
[7] J Gynecol Oncol. 2021 Nov;32(6):e79.
[8] Cancer. 2022 Mar 15;128(6):1206-1218.
[9] Clin Cancer Res. 2021 May 1;27(9):2613-2623.
[10] Int J Gynaecol Obstet. 2022 Sep;158(3):520-527.
[11] Front Oncol. 2021 Feb 26:11:634857.
[12] Gynecol Oncol. 2022 May;165(2):376-384.
[13] J Clin Oncol. 2023 Sep 20;41(27):4369-4380.
[14] J Clin Oncol. 2020 Oct 10;38(29):3388-3397.
[15] Am J Obstet Gynecol. 2021 Apr;224(4):370.e1-370.e13.
The molecular classification of endometrial carcinoma is suitable for all patients diagnosed with endometrial carcinoma, and is mainly used to supplement the morphological diagnosis of histological samples ( including biopsy samples, curettage samples and surgical resection tissues ).
The main clinical significance of classification lies in : ( 1 ) Prompt prognosis, auxiliary FIGO staging ; (2) Auxiliary selection of appropriate surgical procedures ; ( 3 ) Auxiliary selection of appropriate adjuvant therapy ; ( 4 ) Suggesting the efficacy of conservation therapy ; ( 5 ) For patients with recurrence, it is suggested that immunotherapy and targeted therapy are effective ; ( 6 ) Risk of Lynch syndrome.

1. Nucleic Acid Extraction
2. Library Preparation (3.5 hours total time)
3. Sequencing
4. Auto-data Analysis
5. Report





 闽公网安备35021202000745
闽公网安备35021202000745