




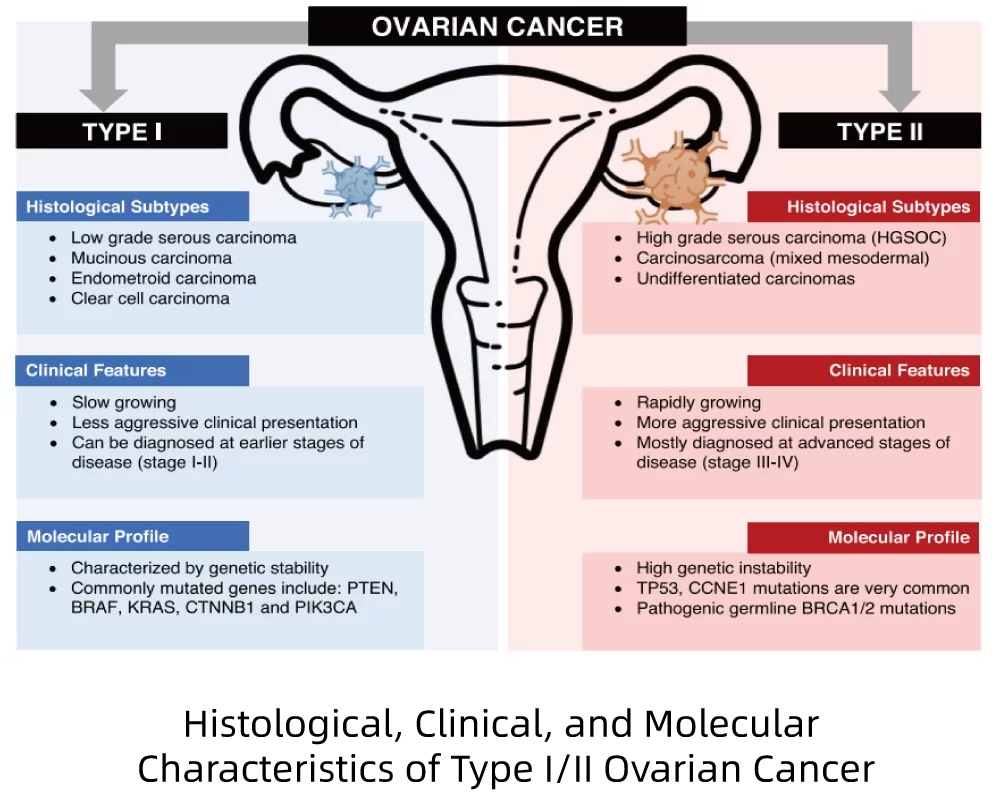
Ovarian cancer has the highest mortality rate among all malignant tumors of the female reproductive tract, with 70% of patients already in the advanced stage at the time of diagnosis. The overall 5-year survival rate ranges from 40% to 50%, while the survival rate for patients with intermediate or advanced-stage disease is approximately 30%. Given that ovarian cancer subtypes exhibit distinct molecular biological mechanisms and prognoses, the corresponding diagnostic tests and treatment strategies should also be tailored accordingly. Immunohistochemistry can effectively support morphological diagnosis; however, the WHO guidelines note that abnormal staining patterns are prone to occur, necessitating adjunctive molecular testing.
In recent years, with the rapid advancement of molecular biology and genetic testing technologies, significant progress has been made in understanding the molecular pathogenic mechanisms of breast cancer. Concurrently, the therapeutic arsenal for breast cancer has been continuously expanded, encompassing endocrine therapy drugs targeting estrogen/progesterone receptors, anti-HER2 agents, PI3K/AKT/mTOR (PAM) signaling pathway inhibitors, anti-angiogenic drugs, PARP inhibitors, CDK4/6 inhibitors, and immune checkpoint inhibitors. Notably, molecular targeted therapy has markedly prolonged the survival of patients with intermediate and advanced-stage breast cancer.
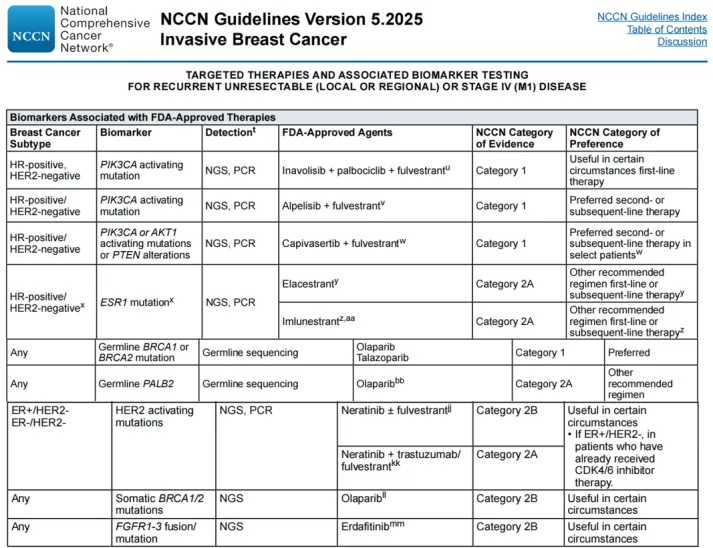
NCCN Guidelines Recommendations for Biomarker Testing
In addition, germline pathogenic variants in the BRCA1/2, TP53, PTEN, and PALB2 genes are associated with an increased risk of breast and ovarian cancer. Therefore, genetic testing for breast and ovarian cancer-associated genes is of great significance for the precise diagnosis and treatment of patients and the improvement of their survival outcomes.
Note: Control samples include whole blood , oral swabs, and saliva.

1. Nucleic Acid Extraction
2. Library Preparation (3.5 hours total time)
3. Sequencing
4. Auto-data Analysis
5. Report




 闽公网安备35021202000745
闽公网安备35021202000745