



The mechanism of tumor occurrence and development is very complex. Although a large number of tumor-related genes have been discovered, there are still many unknown areas of gene action mechanisms, and more scientific research is needed to explore them. Large-scale genetic testing can comprehensively and intuitively characterize tumor genome variation, and benefit to discover rare mutations in more key genes of patients. Through NGS panel detection and matching treatment plans, the overall survival rate of patients is improved, which can provide more treatment opportunities and clinical benefits for patients with advanced tumors.
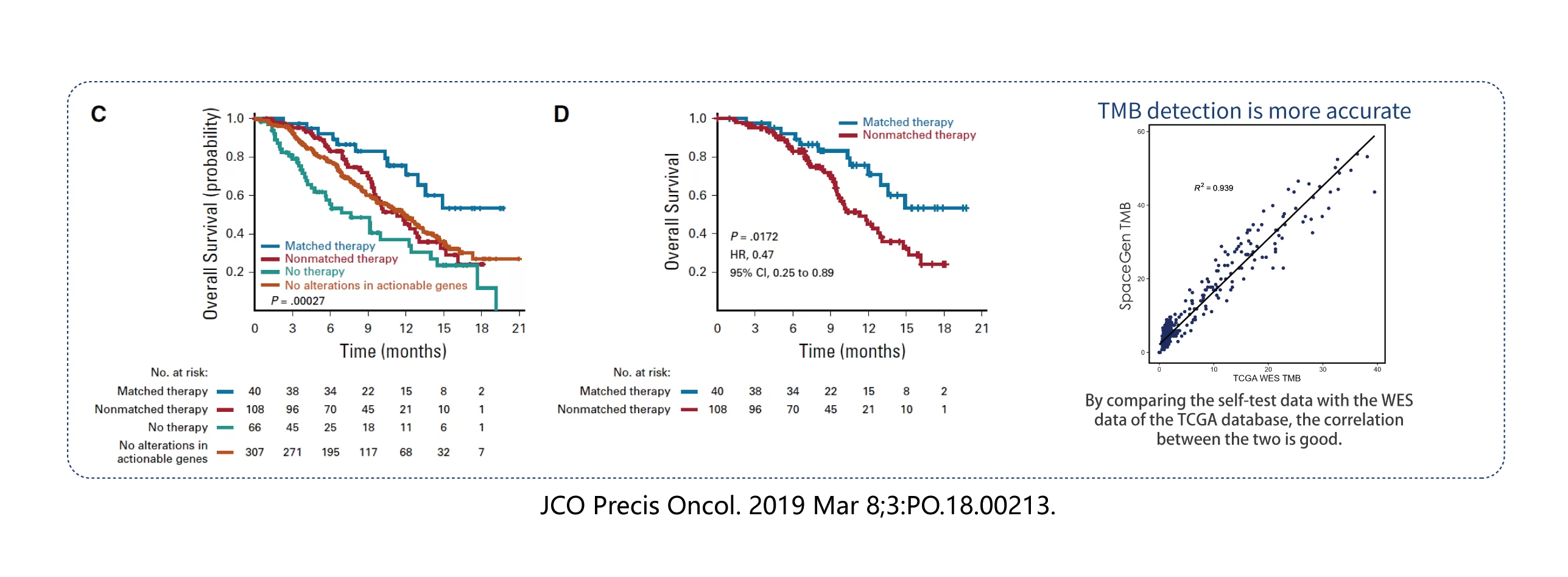
It covers 1200 hot genes in solid tumors, including biomarkers such as MSI, TMB, HRR, HLA, immune resistance, and super-progression, which fully meet the needs of clinical detection.
1、162 targeted drug genes
2、147 genetic related genes
3、164 MSI loci
4、65 immunotherapy-related genes
5、473 signaling pathway-related genes
6、53 chemotherapeutic drug-related sites
7、Tumor Mutation Burden (TMB)
1.The detection of newly diagnosed solid tumor patients before targeted, immune and chemotherapy treatment can assist clinicians in formulating treatment plans and evaluating the prognosis of patients ;
2.Targeted therapy for patients with drug-resistant and chemotherapy-failed solid tumors, new treatment options are needed, genetic testing can be performed to clarify the drug resistance mechanism and find new operable sites ;
3.People with a family history of cancer who need genetic risk assessment ;
4.Patients with solid tumors who can’t obtain pathological tissues and still have the above clinical needs.
Targeted Therapy:Provide comprehensive tumor gene mutation information, including tumor-related pathway gene information and rare sites, analyze tumor pathogenesis, reveal drug resistance mechanisms, and assist clinicians in the rational use of targeted drugs.
Immunotherapy:To comprehensively evaluate the efficacy of immunotherapy, including TMB, MSI , HLA, neoantigen prediction, immunotherapy efficacy and hyperprogression genes.
Chemotherapy:53 chemotherapy-related genes, comprehensively evaluate the efficacy and side effects of chemotherapy, and optimize the chemotherapy regimen.
Genetic Predisposition:Comprehensive assessment of hereditary cancers risk.





 闽公网安备35021202000745
闽公网安备35021202000745