



The International Agency for Research on Cancer ( IARC ) has released the latest statistics on the global cancer burden ( based on data for 2022 ), and once again highlighted the current increasing global cancer burden, which deserves worldwide attention. According to the report, in 2022, there are an estimated 20 million new cancer cases worldwide, and about 9.7 million people died of cancer. It is estimated that the number of survivors within 5 years after cancer diagnosis is about 53.5 million. About 1/5 of people will have cancer in their lives, about 1/9 of men and 1/12 of women died of cancer .It is estimated that by 2050, there will be more than 35 million new cancers worldwide.
Cancer is a complex, multi-gene disease caused by the gradual accumulation of genetic mutations. When genes that regulate cell growth undergo mutations or damage, cells lose control and undergo uncontrolled and unlimited proliferation and differentiation, leading to the development of malignant tumors.
With the advent of precision medicine in cancer treatment, conventional pathological diagnostic methods are no longer sufficient to meet clinical demands. International guidelines and consensuses recognize the value of comprehensive molecular testing based on Next-Generation Sequencing (NGS) in guiding clinical treatment. Comprehensive and accurate tumor genetic diagnostic results have become an essential requirement in clinical diagnosis and treatment.
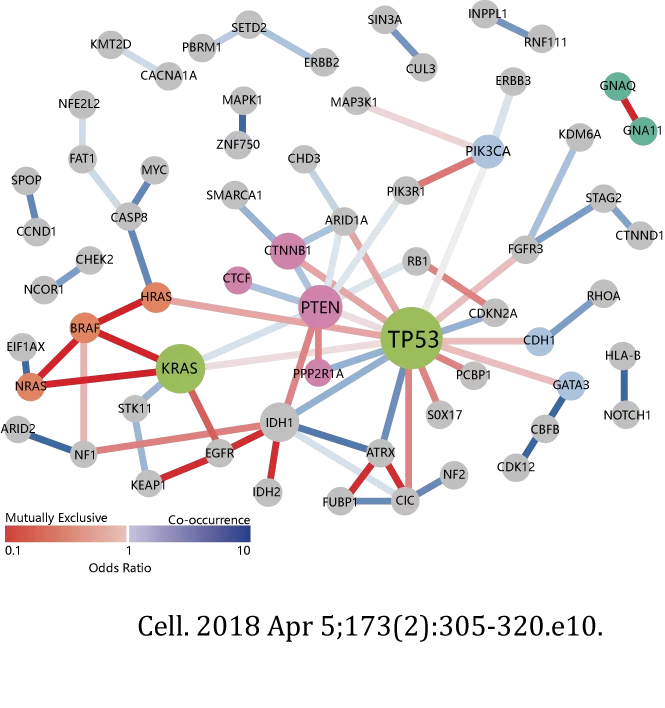
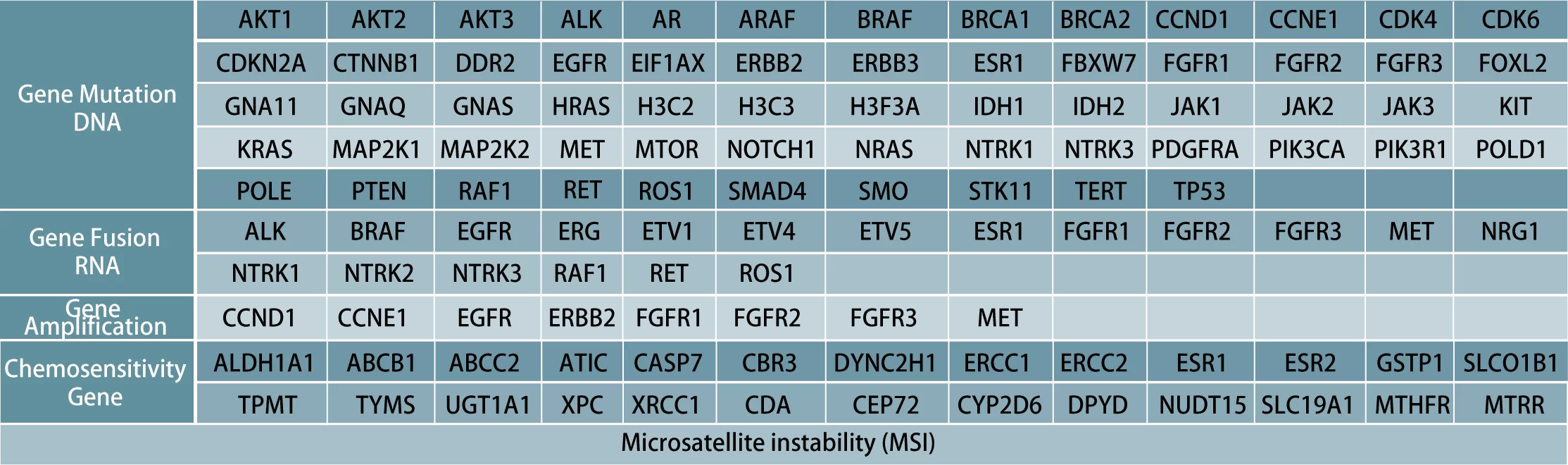
1.The detection of newly diagnosed solid tumor patients before targeted therapy, immunotherapy and chemotherapy can assist clinicians in formulating treatment plans and evaluating the prognosis of patients.;
2.Targeted therapy for patients with drug-resistant and chemotherapy-failed solid tumors requires new treatment options, genetic testing, identification of drug resistance mechanisms, and search for new operable sites.

1. Nucleic Acid Extraction
2. Library Preparation (3.5 hours total time)
3. Sequencing
4. Auto-data Analysis
5. Report





 闽公网安备35021202000745
闽公网安备35021202000745