



The last three decades have witnessed steady, worldwide increases in the incidence of TC . Rates vary widely from country to country, with the highest figures (per 100 000 person-years) reported in Lithuania (15.5), Italy (13.5), Austria (12.4), Croatia (11.4) and Luxembourg (11.1). Estimated TC-related mortality rates, by contrast, are low (0.7 and 0.5 cases per 100 000 person-years for women and men, respectively) with considerably less regional and temporal variation。Thyroid carcinoma occurs two to three times more often in women than in men. Thyroid carcinoma is currently the seventh most common malignancy diagnosed in women.
To understand the pathogenesis of thyroid carcinoma from the genetic level, identify low-risk and high-risk thyroid carcinoma, so as to make a clear diagnosis and scientific individualized management, avoid overtreatment, and establish a precise, standardized and individualized management model.
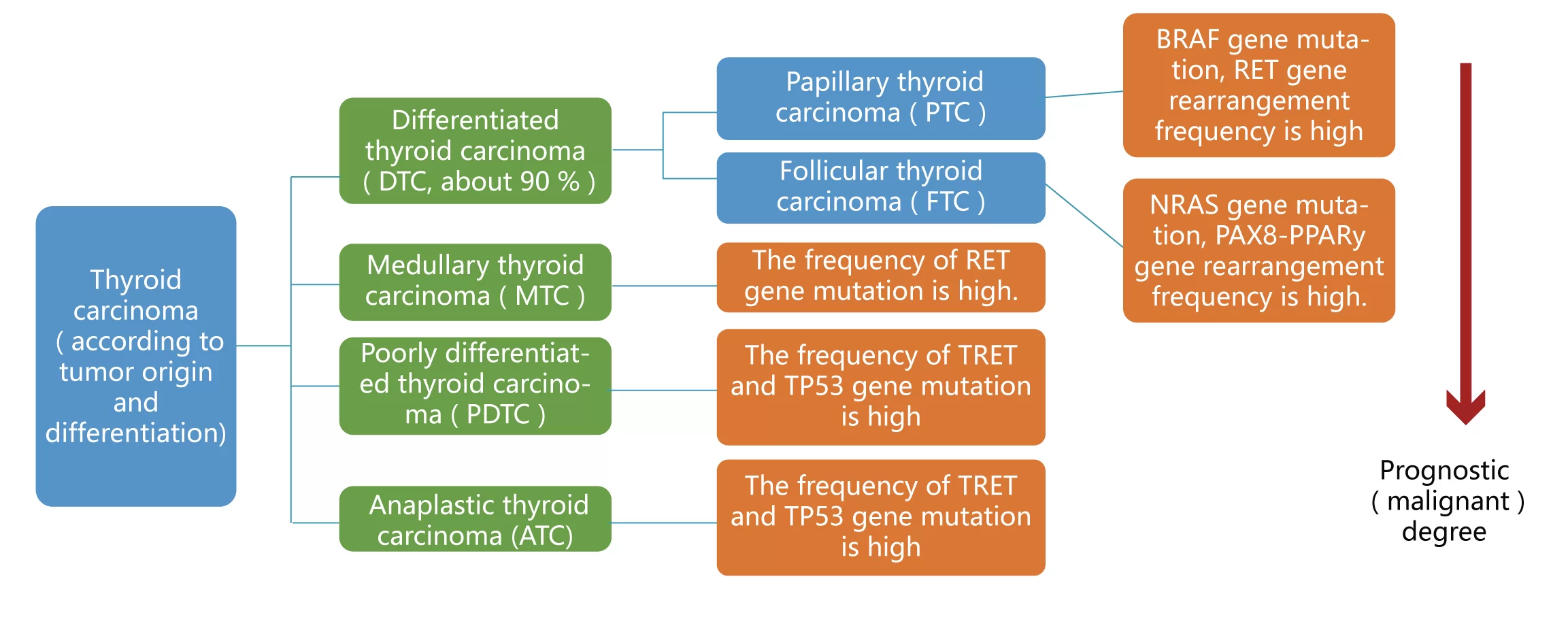
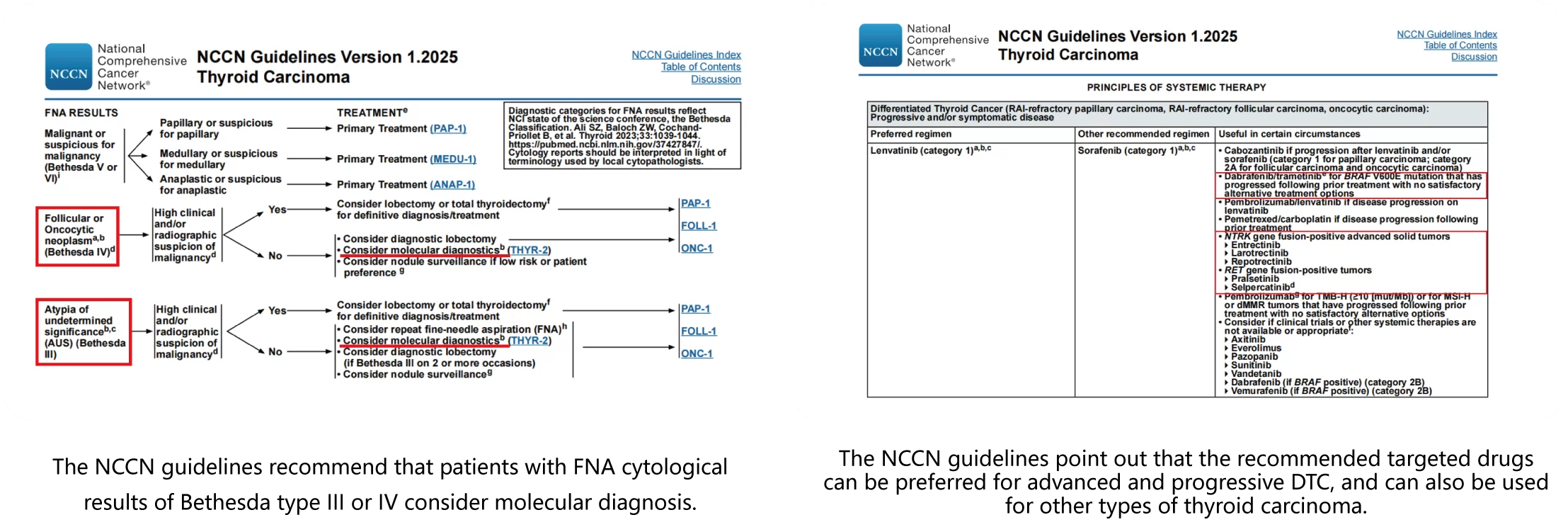
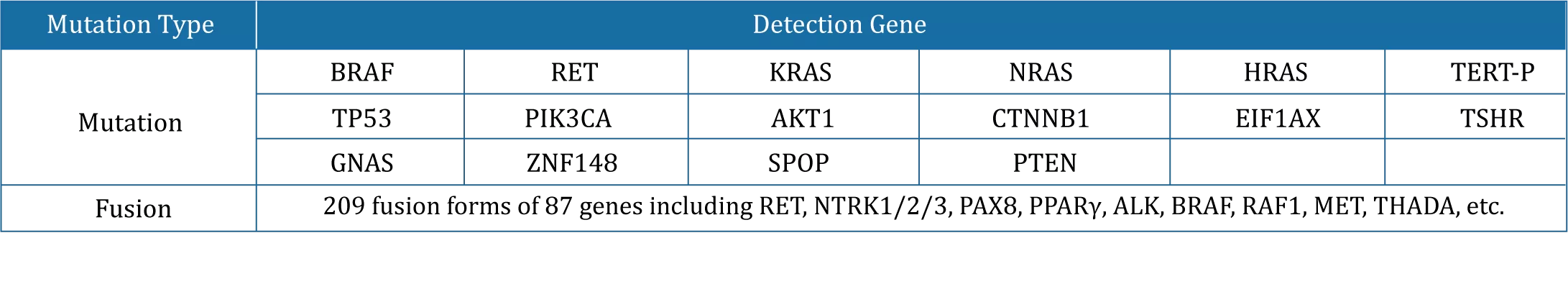
The test kit is based on high-throughput sequencing, covering all common genes related to thyroid tumor typing, prognosis, medication and genetics recommended in NCCN guidelines, CSCO guidelines and expert consensus.
1.Patients with thyroid nodules with uncertain results diagnosed by US-FNA cytology ;
2.Patients undergoing surgical treatment, radioactive iodine therapy or ablation ;
3.Patients with advanced thyroid carcinoma to be selected for targeted therapy ;
4.Patients with hereditary MTC background and their families.
5.Patients with postoperative recurrence.
( 1 ) To assist the diagnosis of benign and malignant thyroid nodules and the classification of thyroid carcinoma.
( 2 ) To guide the choice of surgical plan.
( 3 ) Guidance of radioactive iodine therapy
( 4 ) Guiding targeted therapy
( 5 ) Assessing genetic risk
1. Nucleic Acid Extraction
2. Library Preparation (3.5 hours total time)
3. Sequencing
4. Auto-data Analysis
5. Report






 闽公网安备35021202000745
闽公网安备35021202000745