[2] Front Oncol. 2022 Mar 4:12:856452.
[3] Genes (Basel). 2023 Nov 9;14(11):2060.
[4] Urol Int. 2024 Sep 2:1-5.
[5] Obstet Gynaecol. 2021 Jan;23(1):9-20.
[6] Cancer. 2022 Mar 15;128(6):1206-1218.




Lynch Syndrome (LS) accounts for approximately 2%–4% of all colorectal cancers (CRC) [1]and 3%–5% of all endometrial cancers (EC). LS is primarily caused by germline mutations in mismatch repair (MMR) genes, though rare cases result from germline deletions in the 3' region of the EPCAM gene upstream of MSH2, leading to secondary epigenetic mutations in MSH2[2]. These are associated with deficient mismatch repair (dMMR)/microsatellite instability-high (MSI-H) in tumor tissues. LS patients have a high risk of developing CRC and EC, up to 80% and 60% respectively, and are also at risk for other cancer types such as gastric, small intestine, urothelial, and ovarian cancers[3]. SpaceGen and its partners have also reported cases of LS-associated penile cancer[4]. Guidelines recommend that CRC, EC, and other tumor patients with positive MMR immunohistochemistry (IHC) or MSI screening results, and negative MLH1 methylation or BRAF V600E mutation testing, undergo germline testing for MMR genes to aid in the diagnosis of LS[1].
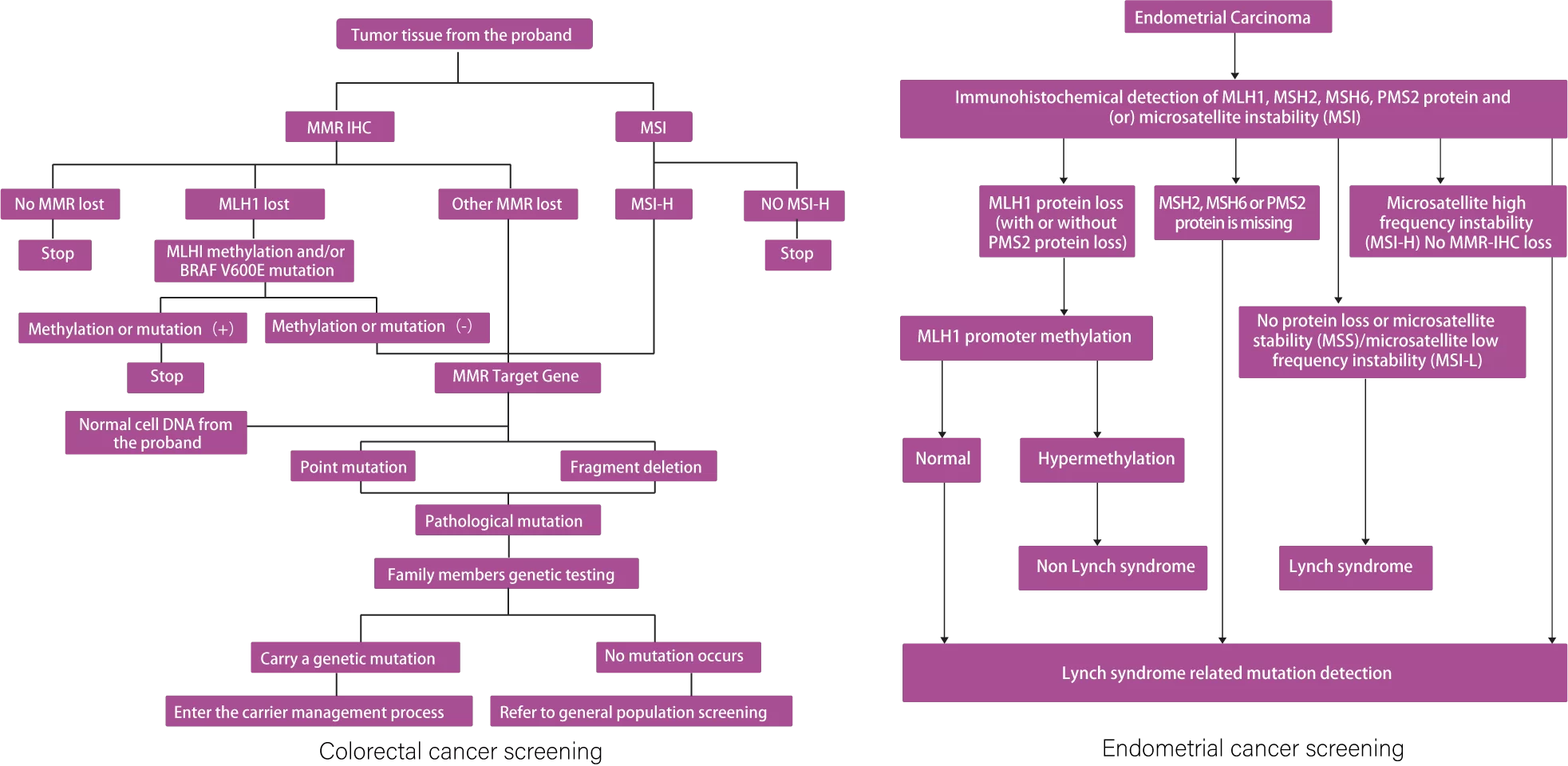
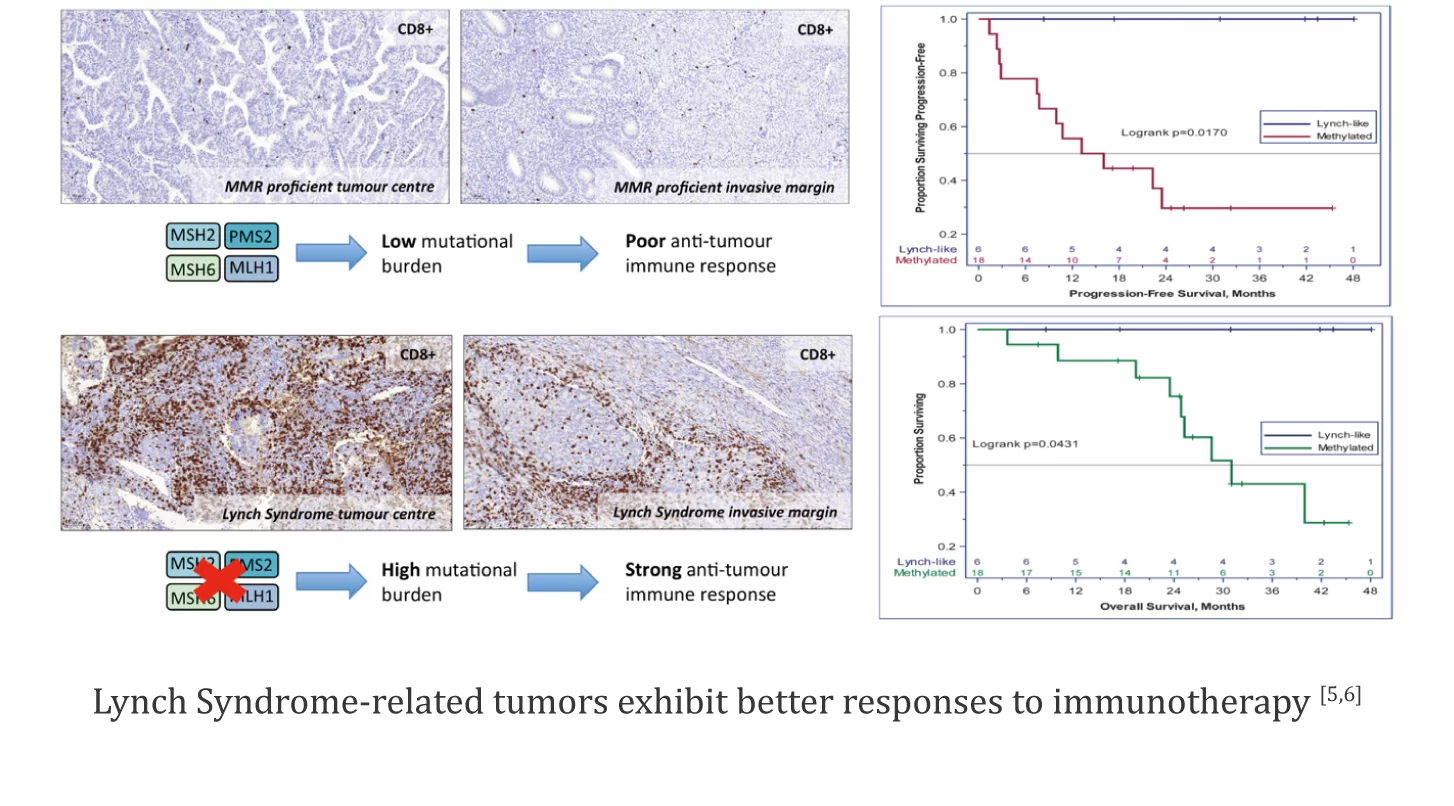
Multiple guidelines and consensus statements indicate that routine MMR/MSI testing combined with BRAF V600E screening may miss 10% of Lynch syndrome (LS) patients, and suggest considering high-throughput sequencing as an alternative to conventional MMR/MSI testing. Additionally, some studies have shown that LS-associated tumors exhibit stronger anti-tumor immune responses compared to sporadic tumors, leading to better efficacy with immune checkpoint inhibitors[5,6].
[2] Front Oncol. 2022 Mar 4:12:856452.
[3] Genes (Basel). 2023 Nov 9;14(11):2060.
[4] Urol Int. 2024 Sep 2:1-5.
[5] Obstet Gynaecol. 2021 Jan;23(1):9-20.
[6] Cancer. 2022 Mar 15;128(6):1206-1218.
Assist in the diagnosis of Lynch syndrome;
Suggest that patients with Lynch-related tumors may benefit from immunotherapy.
1.Patients with CRC, EC, gastric cancer, or other cancer whose tumor tissue tests positive for dMMR/MSI-H, including those negative for MLH1 methylation and BRAF V600E testing;
2.Patients with tumor tissue screening showing proficient mismatch repair (pMMR) or microsatellite stability (MSS) but with high clinical suspicion of Lynch syndrome (LS);
3.Patients with blood-related family members diagnosed with LS;
4.Patients with pathogenic mutations in MMR genes detected in tumor tissue but with unclear germline mutation status;
5.Tissue-based testing may be considered as an alternative to MMR/MSI screening to reduce missed detection of LS.
1. Nucleic Acid Extraction
2. Library Preparation (3.5 hours total time)
3. Sequencing
4. Auto-data Analysis
5. Report