



Over the past 30 years, the incidence of thyroid cancer has continued to rise rapidly in many countries and regions around the world. In 2022, there will be nearly 800,000 new cases worldwide.Genetic diagnosis based on molecular alterations in thyroid cancer has been applied as an auxiliary tool for pathological diagnosis in clinical practice.
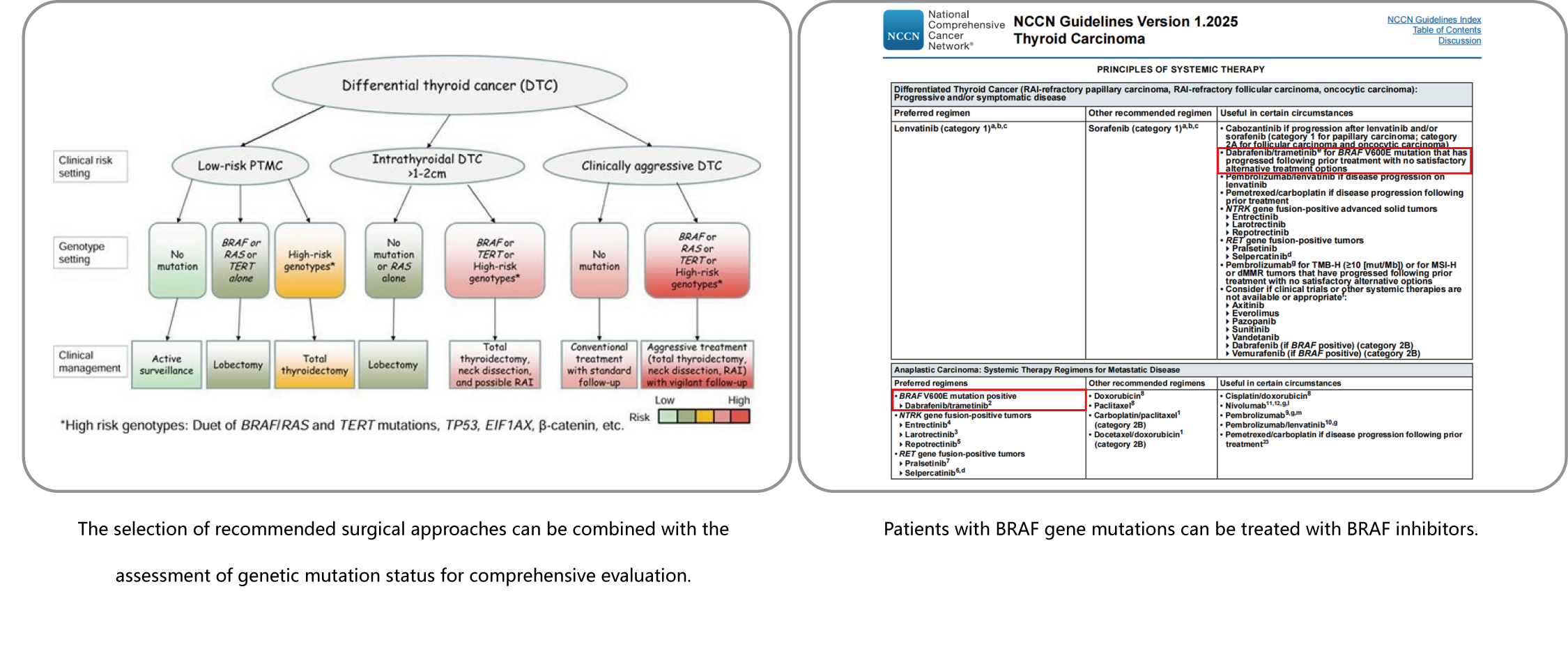
Among the various histological subtypes of thyroid cancer, papillary thyroid carcinoma (PTC) is the most common type. Studies have shown that BRAF is the most frequently mutated gene in PTC, with a mutation frequency of up to 80%, and over 90% of these mutations are the V600E type. This leads to continuous activation of the BRAF gene, which abnormally activates the MAPK signaling pathway and ultimately promotes tumor development. Multiple guidelines and consensus statements indicate that thyroid nodules with BRAF gene mutations carry a 99.8% risk of malignancy and require active treatment.
The TERT gene is also frequently found mutated in PTC, with a mutation frequency of 10% to 15%. However, TERT gene mutations are more common in poorly differentiated thyroid carcinoma (PDTC) and anaplastic thyroid carcinoma (ATC), occurring at a frequency of 40% to 45%. Most TERT gene mutations occur at C228T and C250T in the promoter region, leading to overexpression of telomerase. This disrupts the normal cell proliferation cycle and promotes tumor invasion and metastasis. Clinical guidelines worldwide emphasize that the TERT gene serves as a critical tumor marker for assessing thyroid cancer aggressiveness and predicting poor prognosis. The co-occurrence of TERT and BRAF gene mutations specifically indicates significantly increased tumor aggressiveness and worse prognosis, necessitating aggressive treatment strategies.
1.Patients with thyroid nodules with uncertain results diagnosed by US-FNA cytology ;
2.Patients with advanced thyroid cancer to be selected for targeted therapy ;
3.Patients planned for surgical or ablation therapy.
1. Assist in the diagnosis of benign and malignant thyroid nodules.
2. To guide the choice of surgical plan.
3. To guide targeted therapy
1、Nucleic Acid Extraction
2、Set up qPCR
3、Amplification
4、Data Analysis



 闽公网安备35021202000745
闽公网安备35021202000745