



ALK is an anaplastic lymphoma kinase, and ALK gene fusion mutation is a common driver gene in non-small cell lung cancer (NSCLC), and the proportion of ALK fusion mutation positive in non-small cell lung adenocarcinomas in China is 3-13%. Although the proportion of ALK-positive lung cancer is very low, the number of new cases in China is still close to 35,000 cases per year. Therefore, it is necessary to use genetic testing to accurately detect ALK-positive NSCLC and carry out targeted therapy.
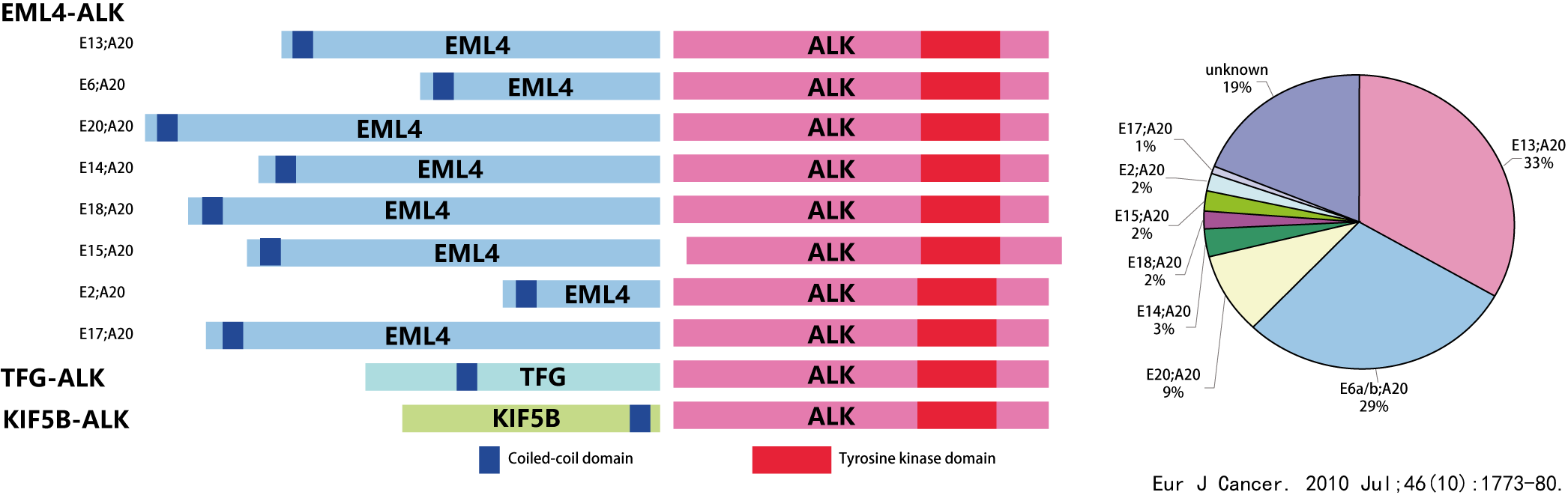
Among the reported ALK fusions, EML4 is the most common fusion partner, which can be divided into different fusion subtypes according to different fracture sites.
Since the approval of Crizotinib in 2011, seven ALK-targeted drugs have been approved for the treatment of advanced ALK+ NSCLC, including Crizotinib, Alectinib, Ceritinib, Ensartinib, Brigatinib, Lorlatinib, Envonalkib and Iruplinalib.
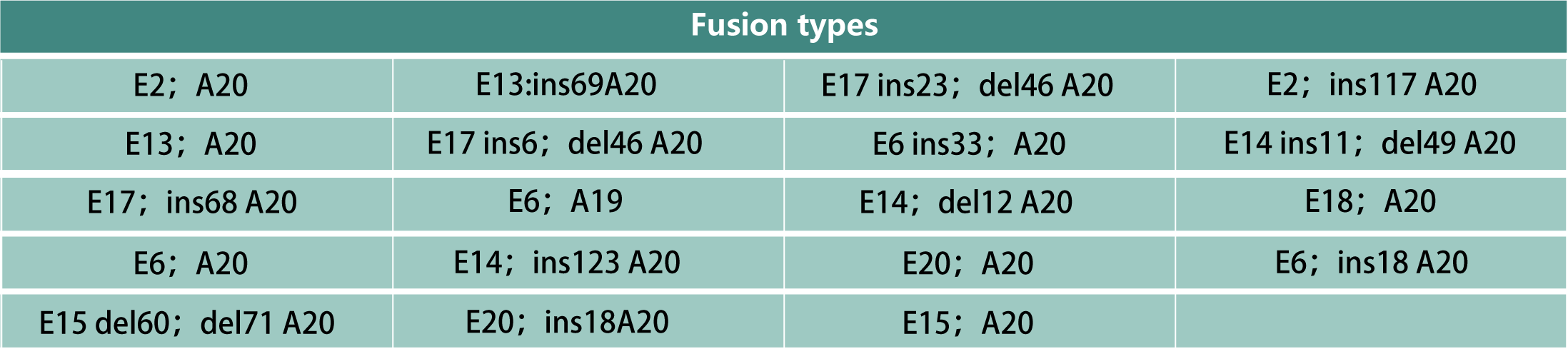
1.Qualitatively detecting EML4-ALK gene fusion mutations in RNA samples assist to evaluate the benefit of treatment with ALK kinase inhibitors.
2.It is suitable for NSCLC patients to use before entering targeted therapy, and provides scientific basis for individual drug use for tumor patients.
1.Nucleic acid extraction
2.Set up qPCR
3.Amplification
4.Data analysis



 闽公网安备35021202000745
闽公网安备35021202000745