



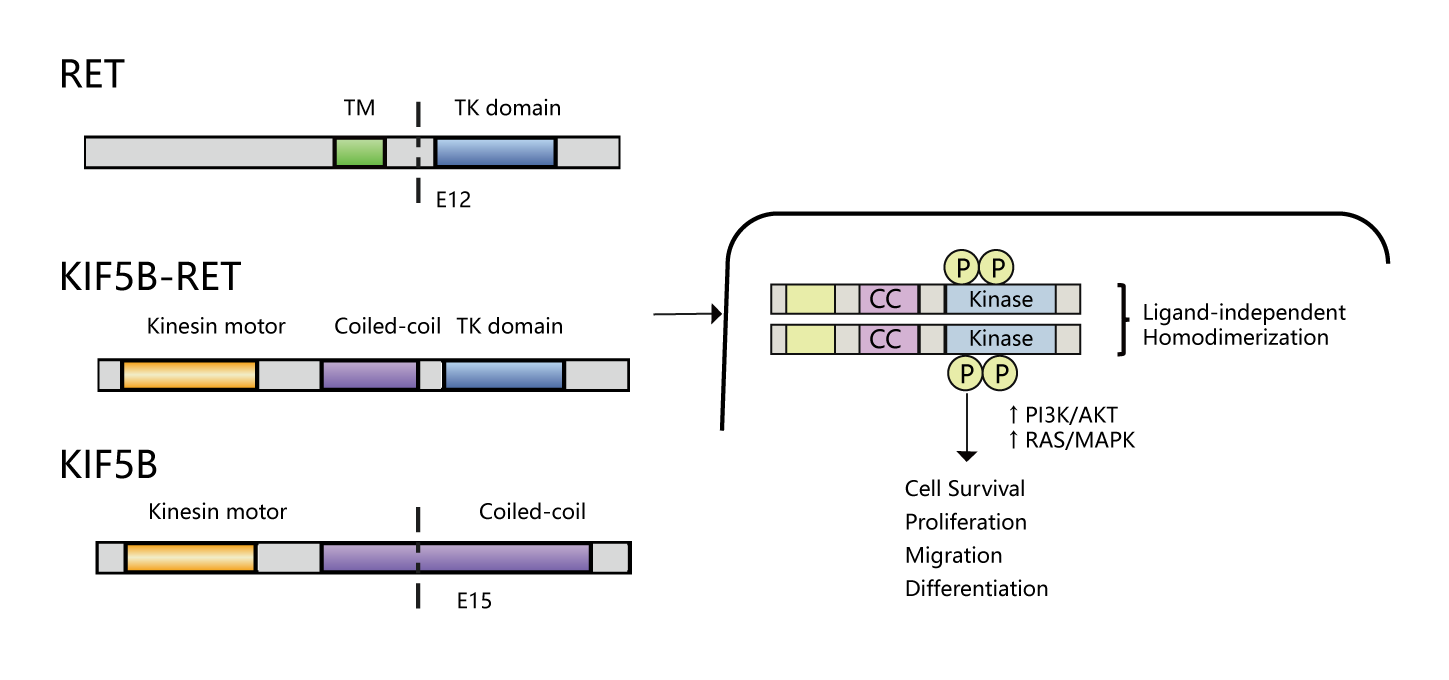
The RET oncogene is located at 10q11.2, 60 kb length and contains 21 exons. After the fusion of RET gene, the dimerization can be completed without ligands, and the RET tyrosine kinase region and downstream signaling pathways such as Ras/Raf/MEK/ERK and PI3K/AKT are continuously activated, which continuously drives the proliferation, migration and differentiation of cells, and thus causes the development of tumors. KIF5B is the most common RET fusion partner in NSCLC[1].
[1] Oncologist. 2013;18(7):865-75.
In 2020, the FDA approved Selpercatinib and Pralsetinib for systemic treatment in patients with RET fusion-positive metastatic non-small cell lung cancer (NSCLC). In March 2021, the NMPA approved Pralsetinib for the treatment of RET fusion-positive adult patients with locally advanced or metastatic NSCLC who have previously received platinum-containing chemotherapy. In September 2022, the RET inhibitor Selpercatinib was approved by NMPA for the treatment of adult patients with RET gene fusion-positive locally advanced or metastatic non-small cell lung cancer (NSCLC).
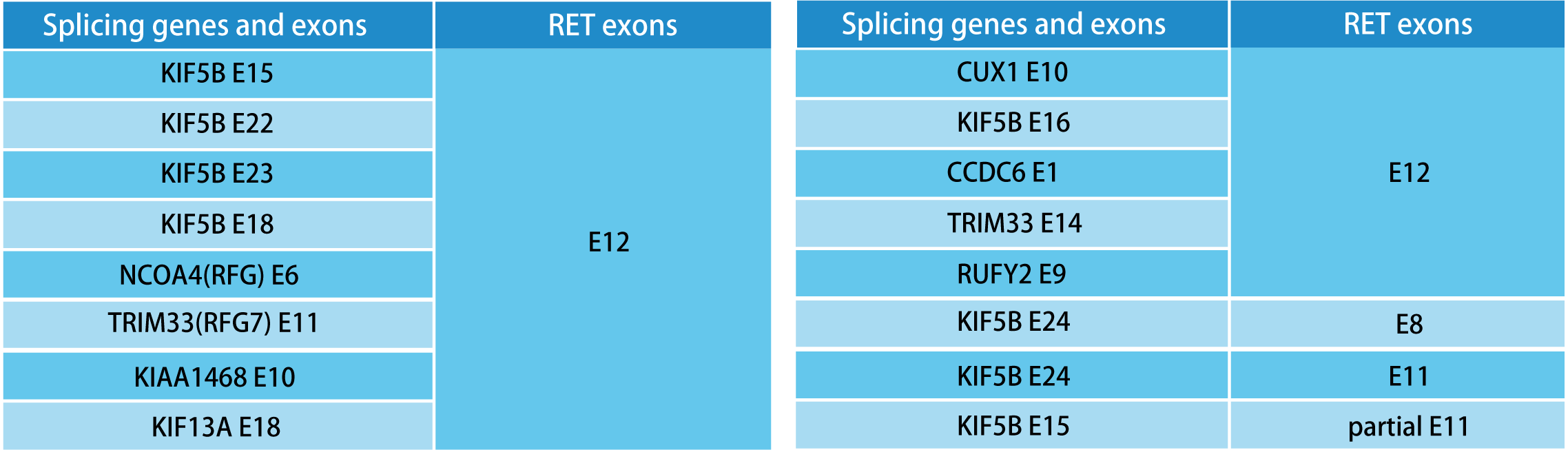
RET gene fusion detection is performed before systematic treatment for inoperable NSCLC patients, and treatment is guided according to molecular classification.
1.Nucleic acid extraction
2.Set up qPCR
3.Amplification
4.Data analysis



 闽公网安备35021202000745
闽公网安备35021202000745