



Lymphatic plasma cell lymphoma (Lympho plasmacytic lymphoma, LPL)/Fahrenheit gigantic globulin hematic disease (Waldenstrom macroglobulinemia, WM) is a rare sample plasma cells differentiation characteristics of small inert B cell lymphoma, The incidence of non-Hodgkin lymphoma is <2%. In recent years, studies have found that more than 90% of LPL/WM patients have Myeloid differentiation factor 88 (MyD88) gene L265P mutation, which is extremely rare in other types of inert B-cell lymphoma, which provides clinical molecular diagnostic basis. The discovery of this recurrent somatic mutation opens a new chapter in the diagnosis, treatment and prognosis assessment of LPL/WM. MYD88 mutations activate the BTK pathway and lead to abnormal activation of the NF-κB signaling pathway, which affects the prognosis of patients. Therefore, BTK inhibitors can inhibit this pathway and have a significant effect on WM patients.
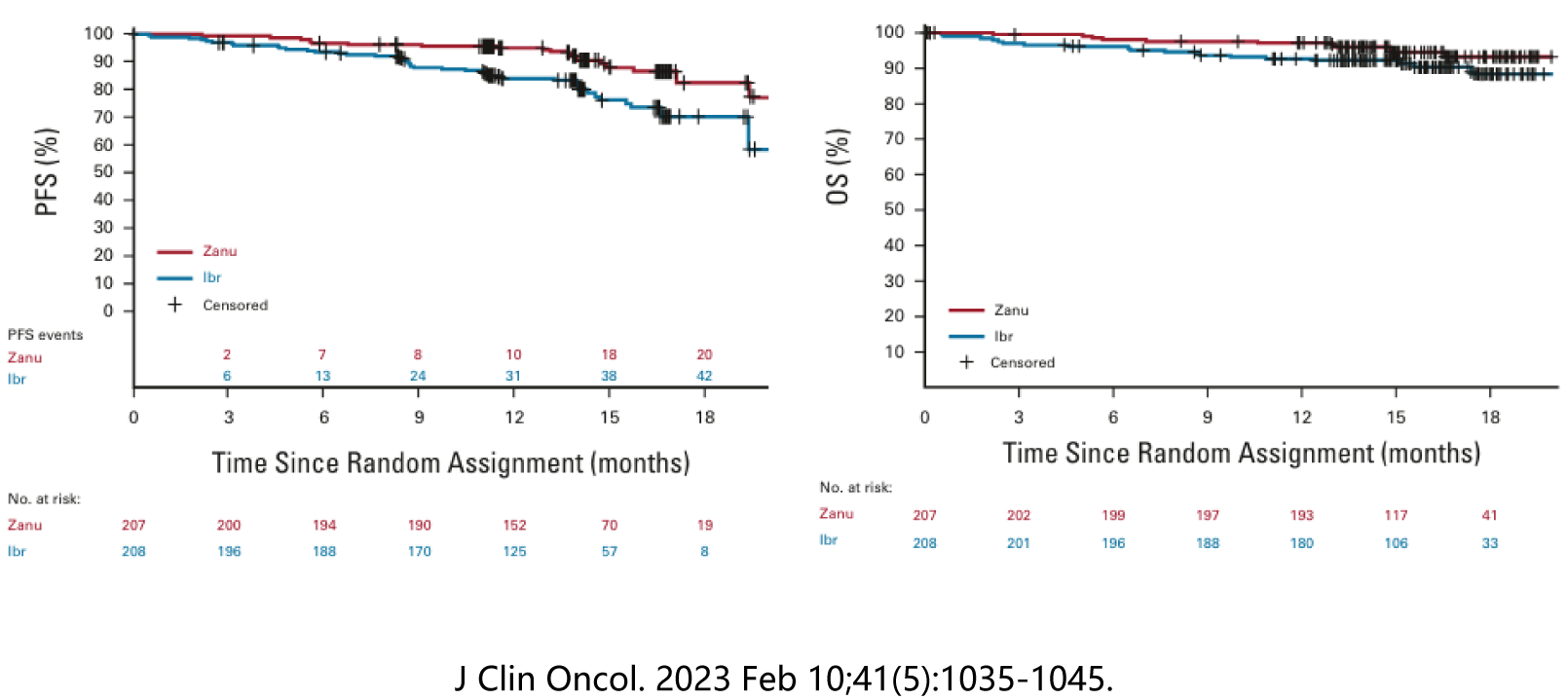
The new generation of BTK inhibitor Zanubrutinib enhances the selectivity of BTK on the basis of the previous BTK inhibitors, reduces the off-target effect, and enhances the curative effect. Zanubrutinib treated wit WM has been listed in the domestic medical insurance market, the domestic accessibility has been greatly enhanced, the economic burden of patients using BTK inhibitor treatment is greatly reduced, and Zanubrutinib also shows a certain price advantage among similar products, so more patients will benefit from the treatment of this drug in the future.
The L265P mutation of the MYD88 gene is detected in the DNA sample.
1. Patients who need assistance in the diagnosis of LPL/WM.
2. Patients with LPL/WM are tested prior to treatment and may be assisted with medication guidance.
1.Nucleic Acid Extraction
2.Set up qPCR
3.Amplification
4.Data Analysis



 闽公网安备35021202000745
闽公网安备35021202000745