



The mutation rates of KRAS gene and NRAS gene in colorectal cancer are about 40 % and 4 %. The RAS / RAF / MAPK pathway is located downstream of EGFR, and the RAS gene is wild type under normal conditions. However, if the pathogenic mutation occurs, the upstream EGFR does not need to receive extracellular signals to automatically activate the signaling pathway, causing excessive cell proliferation. Therefore, patients with wild-type RAS gene are effective for anti-EGFR monoclonal antibody therapy. The guideline consensus recommends the detection of KRAS and NRAS exon 2 / 3 / 4 mutations in all metastatic colorectal cancer.
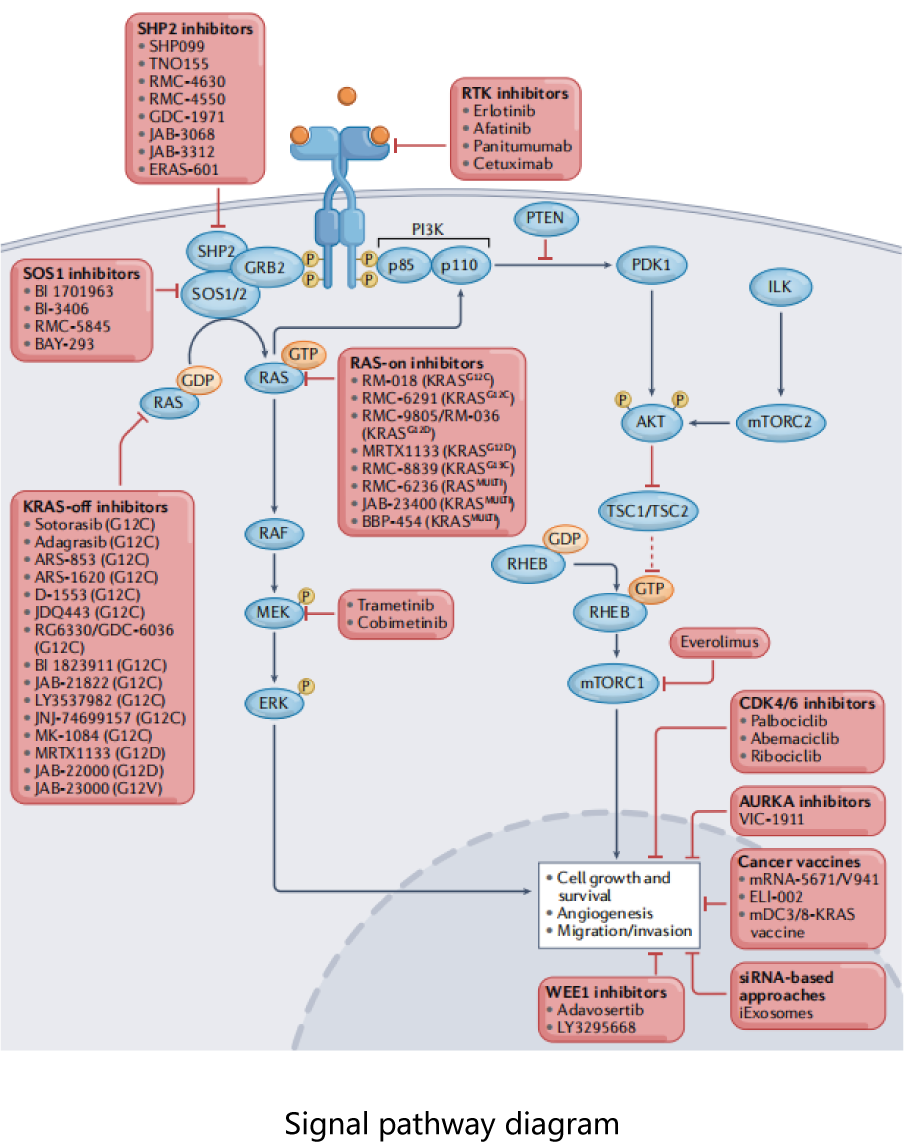
The prognosis of KRAS, NRAS, and BRAF gene mutations is generally poor, and colorectal cancer patients with such mutations are not sensitive to anti-EGFR antibody drugs. The NCCN guidelines recommend that RAS gene status must be identified before treatment of colorectal cancer, and KRAS, NRAS and BRAF genes are recommended for detection when recurrent or metastatic colorectal cancer is identified. According to some existing research results, PIK3CA mutation may be an effective predictive marker for Aspirin .

[1]Nature Reviews Clinical Oncology volume 19, pages637–655 (2022)
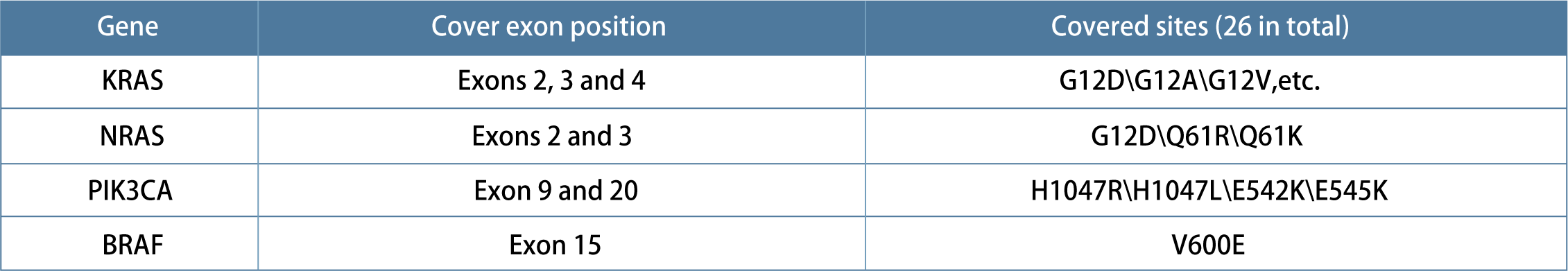
1.The detection of RAS and BRAF gene mutations suggests the use of targeted drugs such as anti-EGFR, BRAF / MEK inhibitors, and HER2 inhibitors. And it has prognostic significance for patients with colorectal cancer.
2.Assist clinicians to screen out relevant tumor targeted drugs.
1、Nucleic Acid Extraction
2、Set up qPCR
3、Amplification
4、Data Analysis



 闽公网安备35021202000745
闽公网安备35021202000745