



Lung cancer is the malignant tumor with the fastest increasing incidence in China in the past 30 years. From the perspective of pathology and treatment, lung cancer can be roughly divided into non-small cell lung cancer (NSCLC) and small cell lung cancer (SCLC), of which non-small cell lung cancer accounts for about 80%–85%. Non-small cell lung cancer, especially lung adenocarcinoma, contains a large number of altered molecular carcinogenic subunits that allow disease prediction and management based on genetic testing results.
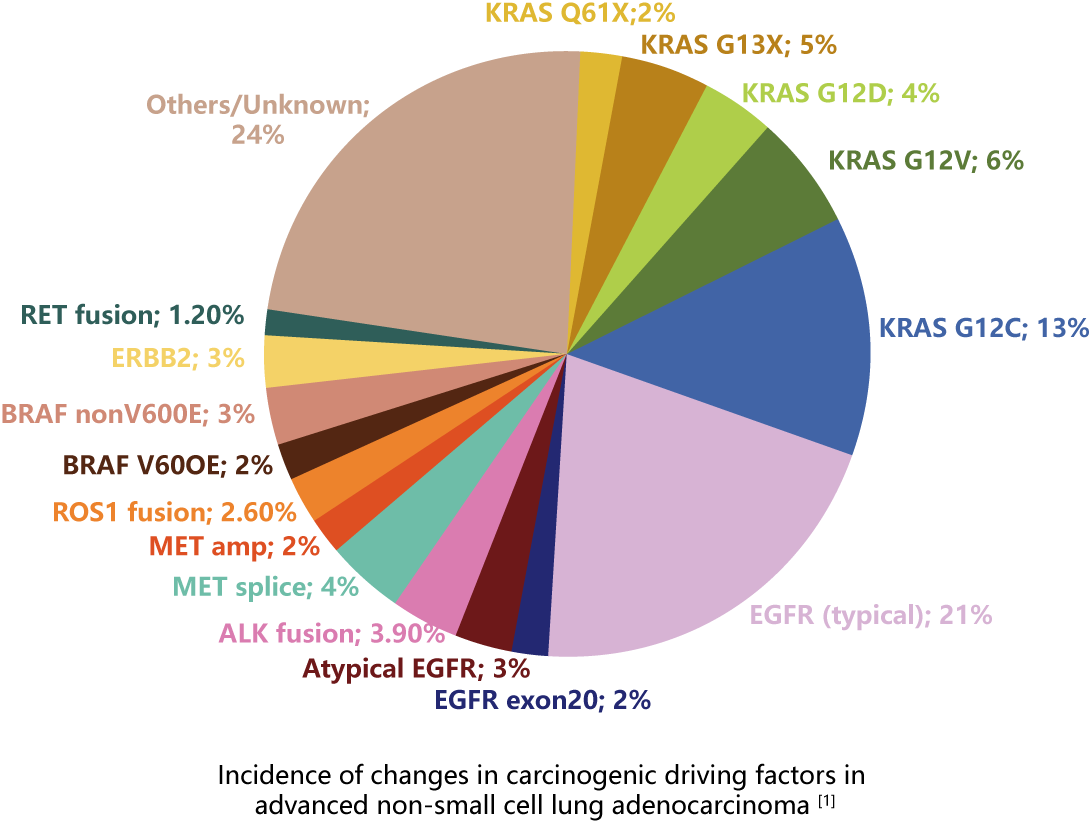
In recent years, with the deepening of research, the treatment of non-small cell lung cancer has achieved great success, especially in the targeted therapy. NCCN guidelines for non-small cell lung cancer clearly state that the mutation status of driver genes is an important factor predicting the efficacy of targeted drug therapy[2]. Patients with human EGFR, KRAS, BRAF, NRAS, HER2 and PIK3CA gene mutations and ALK, ROS1 and RET gene fusion can benefit from the treatment with corresponding tyrosine kinase inhibitors. The mutation status of genes needs to be detected before targeted therapy. The combined detection of multiple gene mutations in patients with non-small cell lung cancer can provide more accurate treatment for patients.

References
[1] Biomark Res.2024 Feb 12; 12(1):24.
[2] NCCN guidelines for non-small cell lung cancer
[3] China Journal of Lung Cancer, 2023, (Issue 11).
Patients with non-small cell lung cancer: especially patients with NSCLC containing an adenocarcinoma component, should undergo genetic testing prior to initial treatment, regardless of their clinical features, in order to provide additional information to the physician in making a treatment decision.
Patients with advanced lung cancer:gene testing can effectively screen people who may benefit from targeted therapy.
For patients with operable NSCLC in stages I to III:postoperative gene testing is recommended to guide adjuvant targeted therapy.
For inoperable patients with NSCLC in stages III to IV:gene testing prior to or during treatment is recommended to guide treatment decisions.
1、Nucleic Acid Extraction
2、Modification
3、Set up qPCR
4、Amplification
5、Data Analysis



 闽公网安备35021202000745
闽公网安备35021202000745